
Concept explainers
(a)
Interpretation:
The numbers of each type of ions within the unit cell of
Concept Introduction:
The unit cell is the basic structurally repeating units of a crystal.
In a crystal of any type,
Each corner atom is shared by 8 unit cells.
Each face-centered atom is shared by 2 unit cells.
One body-centered atom is shared by only 1 unit cell.
(a)
Answer to Problem 11.126QP
The number of
The number of
Explanation of Solution
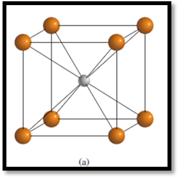
Figure 1
The crystal structure of
The types of ions are:
Determining the number of
The number of
1
One body-centered atom is shared by only 1 unit cell.
The number of
Therefore, the number of
Determining the number of
The number of
8
Each corner atom is shared by 8 unit cells.
The number of
Therefore, the number of
The numbers of each type of ions within the unit cell of
(b)
Interpretation: The numbers of each type of ions within the unit cell of
Concept Introduction:
The unit cell is the basic structurally repeating units of a crystal.
In a crystal of any type,
Each corner atom is shared by 8 unit cells.
Each face-centered atom is shared by 2 unit cells.
One body-centered atom is shared by only 1 unit cell.
(b)
Answer to Problem 11.126QP
The number of
The number of
Explanation of Solution
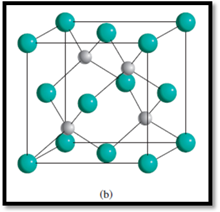
Figure 2
The types of ions are:
Determining the number of
The number of
4
So, the number of
Therefore, the number of
Determining the number of
The number of
8
Each corner atom is shared by 8 unit cells and each face-centered atom is shared by 2 unit cells.
The number of
Therefore, the number of
The numbers of each type of ions within the unit cell of
(c)
Interpretation: The numbers of each type of ions within the unit cell of
Concept Introduction:
The unit cell is the basic structurally repeating units of a crystal.
In a unit cell,
Each corner atom is shared by 8 unit cells.
Each face-centered atom is shared by 2 unit cells.
One body-centered atom is shared by only 1 unit cell.
(c)
Answer to Problem 11.126QP
The number of
The number of
Explanation of Solution
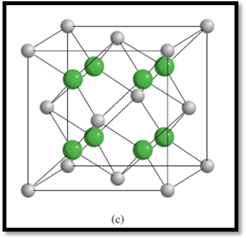
Figure 3
The types of ions are:
Determining the number of
The number of
8
Each corner atom is shared by 8 unit cells and each face-centered atom is shared by 2 unit cells.
The number of
Therefore, the number of
Determining the number of
The number of
8
So, the number of
Therefore, the number of
The numbers of each type of ions within the unit cell of
Want to see more full solutions like this?
Chapter 11 Solutions
AVC LOOSELEAF CHEMISTRY W/CONNECT 2 SEM
- A portion of the crystalline lattice for potassium is illustrated below. (a) In what type of unit cell are the K atoms arranged? A portion of the solid-state structure of potassium. (b) If one edge of the potassium unit cell is 533 pm, what is the density of potassium?arrow_forwardSilicon carbide, SiC, is a very hard, high-melting solid. What kind of crystal forces account for these properties?arrow_forwardCalculate the percent of volume that is actually occupied by spheres in a body-centered cubic lattice of identical spheres You can do this by first relating the radius of a sphere, r, to the length of an edge of a unit cell, l. (Note that the spheres do not touch along an edge but do touch along a diagonal passing through the body-centered sphere.) Then calculate the volume of a unit cell in terms of r. The volume occupied by spheres equals the number of spheres per unit cell times the volume of a sphere (4r3/3).arrow_forward
- What is a lattice? What is a unit cell? Describe a simple cubic unit cell. How many net atoms are contained in a simple cubic unit cell? How is the radius of the atom related to the cube edge length for a simple cubic unit cell? Answer the same questions for the body-centered cubic unit cell and for the face-centered unit cell.arrow_forwardOutline a two-dimensional unit cell for the pattern shown here. If the black squares are labeled A and the white squares are B, what is the simplest formula for a compound based on this pattern?arrow_forwardThe CsCl structure is a simple cubic array of chloride ions with a cesium ion at the center of each cubic array (see Exercise 69). Given that the density of cesium chloride is 3.97 g/cm3, and assuming that the chloride and cesium ions touch along the body diagonal of the cubic unit cell, calculate the distance between the centers of adjacent Cs+ and Cl ions in the solid. Compare this value with the expected distance based on the sizes of the ions. The ionic radius of Cs+ is 169 pm, and the ionic radius of Cl is 181 pm.arrow_forward
- • describe the arrangement of atoms in the common cubic crystal lattices and calculate the packing efficiency for a lattice.arrow_forwardShown below is the cubic unit cell of an ionic compound. Answer the questions by referring to this structure. Be careful to note that some atoms are hidden by those in front. a One of the spheres (red or green) represents a monatomic, metallic ion. The other color sphere represents a monatomic, nonmetal ion. Which spheres probably represent the metal ion? Explain. b How many red spheres are there in the unit cell? How many green ones? c From the information you have, deduce the general formula of the compound, using M for the metallic element and X for the nonmetallic element. What are the formulas of the ions? d Give an example of a compound that might have this structure. Explain why you think this compound might have this structure. Which ion of this compound would be represented by the red spheres?arrow_forwardThe unit cell of silicon carbide, SiC, is illustrated below. (a) In what type of unit cell are the (dark gray) C atoms arranged? (b) If one edge of the silicon carbide unit cell is 436.0 pm, what is the calculated density of this compound? A portion of the solid-state structure of silicon carbide.arrow_forward
- Rutile, TiO2, crystallizes in a structure characteristic of many other ionic compounds How many formula units of TiO2 are in the unit cell illustrated here? (The oxide ions marked by an x are wholly within the cell; the others are in the cell faces.) Unit cell for rufflearrow_forwardCalculate the percent of volume that is actually occupied by spheres in a face-centered cubic lattice of identical spheres. You can do this by first relating the radius of a sphere, r, to the length of an edge of a unit cell, l. (Note that the spheres do not touch along an edge but do touch along the diagonal of a face.) Then calculate the volume of a unit cell in terms of r. The volume occupied by spheres equals the number of spheres per unit cell times the volume of a sphere (4r3/3).arrow_forwardIn the LiCl structure shown in Figure 9.21, the chloride ions form a face-centered cubic unit cell 0.513 nm on an edge. The ionic radius of Cl- is 0.181 nm. (a) Along a cell edge, how much space is between the Cl- ions? (b) Would an Na+ ion (r=0.095nm) fit into this space? a K+ ion (r=0.133nm)?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





