
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11, Problem 11.58SP
The following pseudo-syntheses (guaranteed not to work) exemplify a common conceptual error.
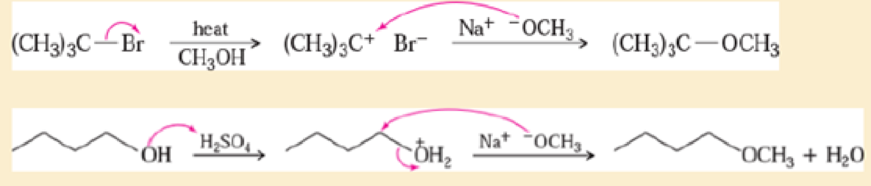
- a. What is the conceptual error implicit in these syntheses?
- b. Propose syntheses that are more likely to succeed.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Provide the missing reagent(s) and major product(s) for the following reactions.
a.
a.ii.
b.
НО.
A + B
a.i.
H₂SO4 (conc.),
A
pyridine
a.iii.
HO
a.iv.
CI
OH
A
B
10. Predict the product for the following reaction.
COH
NHCH,CH,
NHCH,CH3
NCH-CH3
Но
I
II
III
CH,CH,NH,
он
NCH;CH3
IV
V
а. I
b. II
с. II
d. IV
From the table of available reagents select the one(s) you would use to accomplish the transformations shown below.
Reagents Available
a. Crog. H,SO4. H20
b. H2. Pd
c. KMNO4. H20
e. NABH4
f. O; then Zn, CH,CO,H
9. Dess-Martin Periodinane
d. LIAIH,; then H30*
Use the minimum number of steps; in no case are more than two steps necessary.
List reagents by letter in the order that they are used; example: fa.
но-
-CH2OH
OCH3
CO2H
CH2OH
Chapter 11 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 11.1 - Prob. 11.1PCh. 11.2C - Prob. 11.2PCh. 11.3 - Prob. 11.3PCh. 11.3 - Prob. 11.4PCh. 11.3 - Prob. 11.5PCh. 11.3 - Suggest the most appropriate method for each of...Ch. 11.4 - A chronic alcoholic requires a much larger dose of...Ch. 11.4 - Unlike ethylene glycol, propylene glycol...Ch. 11.5 - Predict the major products of the following...Ch. 11.5 - Show how you would convert propan-1-ol to the...
Ch. 11.6 - Predict the products of the following reactions....Ch. 11.7A - Propose a mechanism for the reaction of a....Ch. 11.7B - Prob. 11.13PCh. 11.7B - Show how you would use a simple chemical test to...Ch. 11.7C - Neopentyl alcohol, (CH3)3CCH2OH, reacts with...Ch. 11.7C - Prob. 11.16PCh. 11.7C - When cis-2-methylcyclohexanol reacts with the...Ch. 11.8 - Prob. 11.18PCh. 11.9 - Prob. 11.19PCh. 11.9 - Prob. 11.20PCh. 11.9 - Prob. 11.21PCh. 11.10A - Prob. 11.22PCh. 11.10A - Some alcohols undergo rearrangement or other...Ch. 11.10B - Prob. 11.24PCh. 11.10B - Explain why the acid-catalyzed condensation is a...Ch. 11.10B - Prob. 11.26PCh. 11.10B - When the following substituted cycloheptanol...Ch. 11.11A - Prob. 11.28PCh. 11.11A - Prob. 11.29PCh. 11.11B - Predict the products formed by periodic acid...Ch. 11.12 - Prob. 11.31PCh. 11.13A - Prob. 11.32PCh. 11.14 - Prob. 11.33PCh. 11.14 - a. Show how ethanol and cyclohexanol may be used...Ch. 11.14 - Prob. 11.35PCh. 11.14 - Phenols (pKa 10) are more acidic than other...Ch. 11.14 - To practice working through the early parts of a...Ch. 11.14 - Prob. 11.38PCh. 11 - Predict the major products of the following...Ch. 11 - Show how you would convert 2-methylcyclopentanol...Ch. 11 - In each case, show how you would synthesize the...Ch. 11 - Prob. 11.42SPCh. 11 - Prob. 11.43SPCh. 11 - Prob. 11.44SPCh. 11 - Both cis- and trans-2-methylcyclohexanol undergo...Ch. 11 - Prob. 11.46SPCh. 11 - Prob. 11.47SPCh. 11 - Show how you would make each compound, beginning...Ch. 11 - Predict the major products (including...Ch. 11 - Show how you would use simple chemical tests to...Ch. 11 - The compound shown below has three different types...Ch. 11 - Prob. 11.52SPCh. 11 - Prob. 11.53SPCh. 11 - Prob. 11.54SPCh. 11 - Prob. 11.55SPCh. 11 - Show how you would synthesize the following...Ch. 11 - Show how you would synthesize the following...Ch. 11 - The following pseudo-syntheses (guaranteed not to...Ch. 11 - Two unknowns, X and Y, both having the molecular...Ch. 11 - The Williamson ether synthesis involves the...Ch. 11 - Prob. 11.61SPCh. 11 - Prob. 11.62SPCh. 11 - Alcohols combine with ketones and aldehydes to...Ch. 11 - Prob. 11.64SPCh. 11 - Prob. 11.65SPCh. 11 - Prob. 11.66SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Consider a sample of ideal gas initially in a volume V at temperature T and pressure P. Does the entropy of thi...
General Chemistry: Principles and Modern Applications (11th Edition)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7. Reaction Scheme. NH₂ NH2 or two differnet methods (no same steps/reagents) C5H12N2 1. xs Mel, xs K2CO3 2. Ag2O, H₂O 3. heat Br2, xs NaOH, xs H₂O OHC 1.03 2. DMS CHOarrow_forward1. Predict the products of the following reactions. a. b. MeO C. d. B. Eto 0= O: OEt OMe 1) NaOEt, HOEt 2) CH₂CHCH₂Br 3) NaOH 4) HCI, heat 2) EtBr 3) H₂O* NO₂ + NaOCH, PhCHO PPH3 오 __ NaOH nBuLi CH₂O, HCl(cat) 1) NaOEI, HOEt 2) HCI, H₂O, heatarrow_forwardComplete the following reactions by providing missing reagents or products: i. ii. iii. OH i. ii. H3O+ OEt i. CH3MgCl ii. H3O+ MgCl MgBr i. Xo ii. H3O+ i. NaBH4 ii. H3O+ OHarrow_forward
- I. Predict the outcome of the following reactions. If positive write what will you observe (color, name of the product). If negative, explain the reason why. NH Orcinol но- NH2 A 1.) он NH но NH A 2.) Он ОН %3D HO-P-O-P-0-P-O |3D он (NH4),MoO4 OH OH OH Conc. HNO3 3.) OHarrow_forwarda. CH3CI, AICI3 b. CH3COCI, AICI3 SO₂H Identify the reagents represented by the letters in the above reaction scheme. (Enter your answer as a string of letters without punctuation, e.g. abcd.) Reagents Available c. HNO3, H₂SO4 d. Fe, H3O+ then OH- e. H₂, Pt f. H₂SO4, SO3 Submit Answer b SO3H SO3H SO3H d & ÷ 6 ÷ 6 NH₂ NO₂ NH₂ CI g. Br2, FeBr3 h. Cl2, FeCl3 i. 12, CuCl2 Try Another Version j. KMnO4, H₂O k. NBS, CCl4 1 item attempt remainingarrow_forward2. The following questions relate to the synthesis of cecropia juvenile hormone (this hormone plays an important role in insect development), which is shown below. MECOCH,CO,Et, NaH CO,Me Br CO,Et HO- then H+, heat Meo- OMe RCO3H МеО CO,Me NaH cecropia juvenile hormone a. Suggest reagents to convert 1 into 2 in two steps. b. Provide a reaction mechanism to explain how 2 is converted into 3. Now thinking backwards from the TM (cecropia juvenile hormone), give the structure of compound 5. d. Is the reaction from 5 to the TM regioselective? Explain your answer. с.arrow_forward
- Pick the major product of this reaction. Br لال مع مع O A. I B. || O C. III D. IV O E. All of the above HBr ROOR IIarrow_forward4. Predict the major product for the following reactions. 1. SOCI₂, pyr 2. PhMgBr 3. H a. b. C. d. e. f. 9. NH₂ NH₂ OTS CI 1. H3O*, heat 2. SOCI₂, pyr 1. HNO3, H₂SO4 2. H₂, Pt NH₂ 3. NaNO₂, HCI 4. CuCN 5. H3O*, heat 1. Br₂, FeBr3 2. SOCI₂, pyr. 1. 2 2. xs LAH 3. H₂O 1. HBr, ether 2. NaCN 3. PhMgBr 4. H* 1. NaCN 2. LiAlH4 (xs) 3. H₂O 4. NH₂arrow_forwardAdd arrows indicating the flow of electrons and identify the steps in the mechanism below. H. Ö: H. H. HO: HO: H. H. н H. он HO. H. H. H. H. H. 1) What is purpose of the acid in the first step of the mechanism? 2) How does this help the water react with that intermediate?arrow_forward
- HO A. HO Br B. H3O+ C. Br2, hv The transformations above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order for each transformation, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. HO OF D. E. TBAF OH F. Mg OH OMe (0.5 e Enter correct letters in the correct order to achieve the synthesis in the box below. (Reagents cannot be used more than once.)arrow_forwardIdentify all reactions supported by the substrate below. O a. Sn1 O b.SN2 O C. E1 O d. E2 O e. SN1 & E1 f. SN2 & E2 O8 SN1, SN2 & E1 O h. SN1, E1 & E2 i. SN2, E2 & E1 O j. SN1, SN2, E1 & E2arrow_forward6. Predict the product for the following reaction. "NH₂ Br H₂N NO₂ D. H₂O* A. Fe/HCI IV OH OH Br 11 NH₂ "NH₂ H₂N A. IB. II C. III D. IV E. V 7. Provide the reagents necessary to carry out the following reaction. NH₂ V 1. LIAIH4 2. H₂O NH₂ B. H₂CCOCI C. Br₂/FeBr3 E. dil. Na₂CO3 IIIarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY