
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.10B, Problem 11.27P
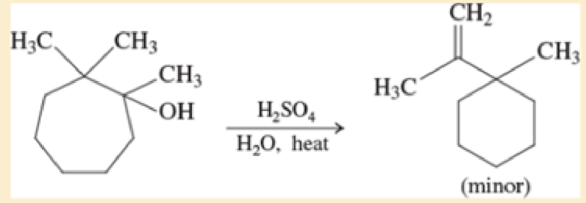
When the following substituted cycloheptanol undergoes dehydration, one of the minor products has undergone a ring contraction. Propose a mechanism to show how this ring contraction occurs.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
propose a mechanism for the following transformations
The Stork reaction is a condensation reaction between an enamine donor and an α,β-unsaturated carbonyl acceptor. The overall reaction consists of a three-step sequence of
formation of an enamine from a ketone,
Michael addition to an α,β-unsaturated carbonyl compound, and
hydrolysis of the enamine in dilute acid to regenerate the ketone.
Consider the Stork reaction between acetophenone and 3-buten-2-one.
Draw the structure of the product of the enamine formed between acetophenone and pyrrolidine.
Draw the structure of the Michael addition product.
Draw the structure of the final product.
Predict the products formed when cyclohexanone reacts with the following reagents. h) sodium acetylide, then mild H3O+
Chapter 11 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 11.1 - Prob. 11.1PCh. 11.2C - Prob. 11.2PCh. 11.3 - Prob. 11.3PCh. 11.3 - Prob. 11.4PCh. 11.3 - Prob. 11.5PCh. 11.3 - Suggest the most appropriate method for each of...Ch. 11.4 - A chronic alcoholic requires a much larger dose of...Ch. 11.4 - Unlike ethylene glycol, propylene glycol...Ch. 11.5 - Predict the major products of the following...Ch. 11.5 - Show how you would convert propan-1-ol to the...
Ch. 11.6 - Predict the products of the following reactions....Ch. 11.7A - Propose a mechanism for the reaction of a....Ch. 11.7B - Prob. 11.13PCh. 11.7B - Show how you would use a simple chemical test to...Ch. 11.7C - Neopentyl alcohol, (CH3)3CCH2OH, reacts with...Ch. 11.7C - Prob. 11.16PCh. 11.7C - When cis-2-methylcyclohexanol reacts with the...Ch. 11.8 - Prob. 11.18PCh. 11.9 - Prob. 11.19PCh. 11.9 - Prob. 11.20PCh. 11.9 - Prob. 11.21PCh. 11.10A - Prob. 11.22PCh. 11.10A - Some alcohols undergo rearrangement or other...Ch. 11.10B - Prob. 11.24PCh. 11.10B - Explain why the acid-catalyzed condensation is a...Ch. 11.10B - Prob. 11.26PCh. 11.10B - When the following substituted cycloheptanol...Ch. 11.11A - Prob. 11.28PCh. 11.11A - Prob. 11.29PCh. 11.11B - Predict the products formed by periodic acid...Ch. 11.12 - Prob. 11.31PCh. 11.13A - Prob. 11.32PCh. 11.14 - Prob. 11.33PCh. 11.14 - a. Show how ethanol and cyclohexanol may be used...Ch. 11.14 - Prob. 11.35PCh. 11.14 - Phenols (pKa 10) are more acidic than other...Ch. 11.14 - To practice working through the early parts of a...Ch. 11.14 - Prob. 11.38PCh. 11 - Predict the major products of the following...Ch. 11 - Show how you would convert 2-methylcyclopentanol...Ch. 11 - In each case, show how you would synthesize the...Ch. 11 - Prob. 11.42SPCh. 11 - Prob. 11.43SPCh. 11 - Prob. 11.44SPCh. 11 - Both cis- and trans-2-methylcyclohexanol undergo...Ch. 11 - Prob. 11.46SPCh. 11 - Prob. 11.47SPCh. 11 - Show how you would make each compound, beginning...Ch. 11 - Predict the major products (including...Ch. 11 - Show how you would use simple chemical tests to...Ch. 11 - The compound shown below has three different types...Ch. 11 - Prob. 11.52SPCh. 11 - Prob. 11.53SPCh. 11 - Prob. 11.54SPCh. 11 - Prob. 11.55SPCh. 11 - Show how you would synthesize the following...Ch. 11 - Show how you would synthesize the following...Ch. 11 - The following pseudo-syntheses (guaranteed not to...Ch. 11 - Two unknowns, X and Y, both having the molecular...Ch. 11 - The Williamson ether synthesis involves the...Ch. 11 - Prob. 11.61SPCh. 11 - Prob. 11.62SPCh. 11 - Alcohols combine with ketones and aldehydes to...Ch. 11 - Prob. 11.64SPCh. 11 - Prob. 11.65SPCh. 11 - Prob. 11.66SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Consider a sample of ideal gas initially in a volume V at temperature T and pressure P. Does the entropy of thi...
General Chemistry: Principles and Modern Applications (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals inlarge doses, paraldehyde is used as a sedative or hypnotic. Propose a mechanism for the formation of paraldehyde.arrow_forwardThe Stork reaction is a condensation reaction between an enamine donor and an α,β-unsaturated carbonyl acceptor. The overall reaction consists of a three-step sequence of formation of an enamine from a ketone, Michael addition to an α,β-unsaturated carbonyl compound, and hydrolysis of the enamine in dilute acid to regenerate the ketone. Consider the Stork reaction between cyclohexanone and propenal Draw the structure of the product of the enamine formed between cyclohexanone and dimethylamine. - Michael addition to an α,β-unsaturated carbonyl compound, and - hydrolysis of the enamine in dilute acid to regenerate the ketone.arrow_forwardWhen the ketone and aldehyde shown below are treated with a base two possible products are formed. Discuss how each product is formed and provide a mechanism.arrow_forward
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardWhen cyclohexanone is heated in the presence of a large amount of acetone cyanohydrin and a small amount of base, cyclohexanone cyanohydrin and acetone are formed. Propose a mechanism.arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
- Using your reaction roadmap as a guide, show how to convert cyclohexanol into racemic trans-1,2-cydohexanediol. Show all required reagents and all molecules synthesized along the way.arrow_forwardWhat reagents are needed to carry out the following syntheses?arrow_forwardMustard gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagentarrow_forward
- Acetic acid has been mixed with isoamyl alcohol to produce isoamyl acetate giving off a banana smell. Propose a reaction mechanism for this reaction.arrow_forwardWhen the following seven-membered ring alcohol is dehydrated, three alkenes are formed. Propose a mechanism for their formation.arrow_forwardA vicinal diol has OH groups on adjacent carbons. The dehydration of a vicinal diol is accompanied by a rearrangement called the pinacol rearrangement. Propose a mechanism for this reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License