
(a)
Interpretation:
The balanced equation to represent the complete combustion of the given compound is to be stated.
Concept introduction:
Alkane is made of only carbon and hydrogen atoms. These atoms form a bond with each other and with other atoms through the sharing of electrons. They do not completely donate their electrons to other atoms while forming a bond. A general complete combustion reaction of
Answer to Problem 11.60E
The balanced equation to represent the complete combustion of the given compound is shown below as,
Explanation of Solution
The given molecule is butane. The chemical formula of butane is
A general complete combustion reaction of alkanes is shown below as,
Where,
•
The number of carbon atoms in butane is four.
Therefore, the balanced equation to represent the complete combustion of the given compound is shown below as,
The reaction is further simplified as,
The balanced equation to represent the complete combustion of the given compound is shown below as,
(b)
Interpretation:
The balanced equation to represent the complete combustion of the given compound is to be stated.
Concept introduction:
Alkane is made of only carbon and hydrogen atoms. These atoms form a bond with each other and with other atoms through the sharing of electrons. They do not completely donate their electrons to other atoms while forming a bond. A general complete combustion reaction of alkanes is shown below as,
Answer to Problem 11.60E
The balanced equation to represent the complete combustion of the given compound is shown below as,
Explanation of Solution
The given molecule is shown below as,
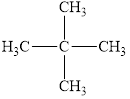
Figure 1
The chemical formula of the given compound is
A general complete combustion reaction of alkanes is shown below as,
Where,
•
The number of carbon atoms in the given compound is five.
Therefore, the balanced equation to represent the complete combustion of the given compound is shown below as,
The balanced equation to represent the complete combustion of the given compound is shown below as,
(c)
Interpretation:
The balanced equation to represent the complete combustion of the given compound is to be stated.
Concept introduction:
Alkane is made of only carbon and hydrogen atoms. These atoms form a bond with each other and with other atoms through the sharing of electrons. They do not completely donate their electrons to other atoms while forming a bond. A general complete combustion reaction of cycloalkanes is shown below as,
Answer to Problem 11.60E
The balanced equation to represent the complete combustion of the given compound is shown below as,
Explanation of Solution
The given molecule is shown below as,
![]()
Figure 2
The chemical formula of the given compound is
A general complete combustion reaction of cycloalkanes is shown below as,
Where,
•
The number of carbon atoms in the given compound is four.
Therefore, the balanced equation to represent the complete combustion of the given compound is shown below as,
The balanced equation to represent the complete combustion of the given compound is shown below as,
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- Draw structural formulas for organic products A and Barrow_forwardDraw the structural formulas corresponding to each of the following IUPAC names: a.ethylcyclobutane b.1, 1, 2, 5-tetramethylcyclohexane c.1-butyl-3-isopropylcyclopentanearrow_forwardHydrocarbons are generally polar. They are generally soluble in organic solvents. a. if the first statement is true and the second statement is false. b. If the first statement is false and the second statement is true. c. If both statements are false. d. If both statements are true.arrow_forward
- A sample petrol gave 85% of carbon and 15% of hydrogen gas. Calculate the weigth of air required for complete combustion of 1kg of petrol.arrow_forwardOrganic Compound: Isooctane A. Give the use of the organic compound in everyday life B. Effects to humans and other living things C. Precautions in using the compoundarrow_forwardVanillin can be recrystallized from water because it is slightly soluble in warm water. Which functional group of vanillin is mostly responsible for the slight water solubility? Why is vanillin not completely soluble in water?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





