
Concept explainers
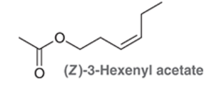
The compound
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEMISTRY,SOLN.MAN.+...-ACCESS
- The following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardAlcohols undergo an oxidation reaction to yield carbonyl compounds on treatment with CrO3. For example, 2-tert-butylcyclohexanol gives 2-tert-butylcyclohexanone. If axial OH groups are generally more reactive than their equatorial isomers, which do you think reacts faster, the cis isomer of 2-tert-butylcyclohexanol or the trans isomer? Explain.arrow_forwardUsing your reaction roadmap as a guide, show how to convert ethylene into 1-butene. All of the carbon atoms of the target molecule must be derived from ethylene. Show all intermediate molecules synthesized along the way.arrow_forward
- Describe how 1-ethylcyclohexanol can be prepared from cyclohexane. You can use any inorganic reagents, any solvents, and any organic reagents as long as they contain no more than two carbons.arrow_forwardThe southern pine beetle utilizes a multi-component aggregation pheromone (one component shown below) to start mass colonization of healthy trees. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. H3C- OH ✔arrow_forward5 Ethanal, CH₂CHO, can be converted by a two-step synthesis into 2-hydroxypropanoic acid. OH O H₂C- H O-H 2-hydroxypropanoic acid (1) The reagents and conditions are 1st step 2nd step NaOH(aq), heat under reflux A Na₂Cr₂O, and dilute H₂SO4 heat under reflux B Cl₂, UV light NaOH(aq), heat und reflux CLIAIH in dry ether CO₂, room temperature D HCN, in presence of KCN(aq) dilute HCl(aq), heat under refluxarrow_forward
- Predict whether each reaction proceeds predominantly by substitution (SN1 or SN2) or elimination (E1 or E2) or whether the two compete. Write structural formulas for the major organic product(s).arrow_forwardThe southern pine beetle utilizes a multi-component aggregation pheromone (one component shown below) to start mass colonization of healthy trees. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. H3C O- 2 3 OH O Proble Atoms, Bo and Ring HS 5 466 HO Sarrow_forwardThe southern pine beetle utilizes a multi-component aggregation pheromone (one component shown below) to start mass colonization of healthy trees. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. HgC O OH aarrow_forward
- Draw the line-angle formula of the enol formed in the following alkyne hydration reaction and then draw the structural formula of the carbonyl compound with which this enol is in equilibrium. -C=CH 1. (sia)2BH 2. NaOH/H₂O₂ an enol carbonyl compound • You do not have to consider stereochemistry. • Draw both the enol and the carbonyl forms. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate structures using the sign from the drop-down menu. ChemDoodleⓇ <arrow_forwardshow-all-working-explaining-detailly-each-step Answer should be typewritten with a computer keyboard.arrow_forwardWhen 2-bromo-2-methylbutane is treated with a base, a mixture of 2-methyl-2-butene and 2-methyl-1- butene is produced. When potassium hydroxide is the base, 2-methyl-1-butene accounts for 45% of the product mixture. However, when potassium tert-butoxide is the base, 2-methyl-1-butene accounts for 70% of the product mixture. What percent of 2-methyl-1-butene would be in the mixture if potassium propoxide were the base? base Br A. Less than 45% B. C. 45% Between 45% and 70% D. More than 70%arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning



