
Concept explainers
Interpretation:
The element that tend to have largest atomic radius in the period should be determined from the given periodic table.
Concept introduction:
A tabular arrangement of the chemical elements by their
Answer to Problem 7STP
The correct option is (c), Y.
Explanation of Solution
Given:
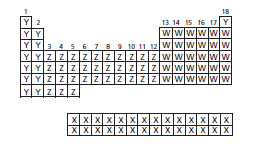
Distance from the nucleus to the valence shell of an atom is said to be its atomic size.
The principal energy level for the elements in the same periods remains same. When moving across the period that is from left to right in the periodic table, addition of one more proton to the nucleus of an atom takes place which results in the increase of nuclear charge by one unit for each step from left to right. Due to increase in nuclear charge, the nucleus of an atom exerts an increasingly strong pull on the electrons present in the valence shell that is valence electrons and thus, atomic radius decreases.
On moving down the group, the electrons are added to higher energy levels and there are more electrons present between nucleus and the valence electrons. Thus, the valence electrons do not feel more attraction towards nucleus. Hence, on moving down the group the atomic size increase.
Hence, the element that tend to have largest atomic radius will be that element which is present at the left side of the periodic table that is, element Y.
Chapter 11 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Organic Chemistry (9th Edition)
CHEMISTRY-TEXT
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach (4th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





