
Concept explainers
Interpretation:
Whether the given reaction is complete or not needs to be determined. Also, the percent yield of the reaction needs to be calculated.
Concept introduction:
Percent yield is the measure of efficiency of the
Explanation of Solution
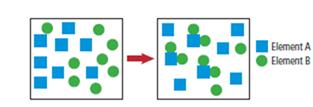
No, reaction is not complete as seen in figure reactants in right hand side.
From figure one can get one atom of A reacting with two atoms of B.
So,
Here, one can see that B is limiting reactant so reaction yield depends on the B,
From stoichiometry of reaction, 2 atoms of B react to produce 1 molecule of AB2
To determine theoretical yield, 8 atoms of B are given. So, it should produce the following molecules of AB2.
Theoretical yield is 4 molecules of AB2
Actual yield given in figure
That is 3 molecules of AB2
Percent yield is the efficiency of a chemical reaction. It is determined using the following relation.
Chapter 11 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
CHEMISTRY-TEXT
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: Structure and Properties (2nd Edition)
Essential Organic Chemistry (3rd Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: A Molecular Approach (4th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





