
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
13th Edition
ISBN: 9780137444298
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.5, Problem 11.24PP
Identify the following as
a.
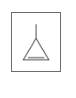
b.
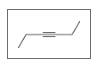
c.
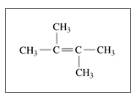
d.
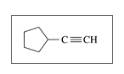
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
write the formula of thefollowing alkanes:a. heptane b. Nonane c. Octane d. Dodecanee. Undecane
Alkenes decolorize bromine water and alkanes do not. Why?
в propyne
2-methyl-2-hexene
HW: Classify each of the following condensed structural or skeletal
f the
formulas as an alkane, an alkene, or an alkyne:
b.
a. CH3-C=C-CH3
Chapter 11 Solutions
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Prob. 11.5PPCh. 11.1 - Prob. 11.6PPCh. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Draw the condensed structural formula for alkanes...Ch. 11.2 - Draw the condensed structural formula for alkanes...
Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Give the IUPAC name for each of the following a....Ch. 11.3 - Give the TUPAC name for each of the following: a....Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the line-angle formula for each of the...Ch. 11.3 - Prob. 11.18PPCh. 11.4 - Heptane, used as a solvent for rubber cement, has...Ch. 11.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Draw the condensed structural formula, or...Ch. 11.5 - Prob. 11.28PPCh. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Draw the condensed structural formula for each of...Ch. 11.6 - Prob. 11.32PPCh. 11.7 - Draw the structural formula for the product in...Ch. 11.7 - Draw the structural formula for the product in...Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11 - Prob. 11.41UTCCh. 11 - Prob. 11.42UTCCh. 11 - Prob. 11.43UTCCh. 11 - Prob. 11.44UTCCh. 11 - Convert each of the following line-angle formulas...Ch. 11 - Convert each of the following line-angle formulas...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name (including cis or trans, if...Ch. 11 - Give the LUPAC name (including cis or trans, if...Ch. 11 - Prob. 11.51APPCh. 11 - Prob. 11.52APPCh. 11 - Name each of the following aromatic compounds:...Ch. 11 - Prob. 11.54APPCh. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Prob. 11.59APPCh. 11 - Draw the line-angle formula for each of the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Give the name for the product from the...Ch. 11 - Give the name for the product from the...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Prob. 11.67CPCh. 11 - Prob. 11.68CPCh. 11 - Prob. 11.69CPCh. 11 - Prob. 11.70CPCh. 11 - Prob. 11.71CPCh. 11 - Prob. 11.72CPCh. 11 - Prob. 11.73CPCh. 11 - Margarines are produced from the hydrogenation of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6. Any organic compound that contains a benzene ring or similar feature. O a. addition reaction Ob. aliphatic compound Oc. alkene d. alkyne Oe. aromatic hydrocarbon Of. hydration g. hydrogenation h. monomer Oi. phenyl group j. polycyclic aromatic hydrocarbon k. polymer I. unsaturated hydrocarbonarrow_forward5. Give the similarities and differences between alkanes, alkenes and alkynes.arrow_forward. Give an example of an Alkane, Alkyne, and Alkene for the following amounts of Carbon using the correct formulas (write each out){ uborg of zeg animus b a. 4 b. 8 wolt villidulos srit novi bang antwollal sr.arrow_forward
- Classify each of the following hydrocarbons as an: alkane, alkene, alkyne or arene (aromatic). H CEC -H H. H- H- H. H. H. H. H. H- alkane v H. H H. H. H.arrow_forwardDraw skeletal structures for the following: a. 5-ethyl-2-methyloctane b. 1,3-dimethylcyclohexane c. 2,3,3,4-tetramethylheptane d. propylcyclopentane e. 2-methyl-4-(1-methylethyl)octane f. 2,6-dimethyl-4-(2-methylpropyl)decanearrow_forwardWhat is the general formula for a noncyclic alkane? C?H a. b. If a noncyclic alkane contains 15 carbon atoms, how many hydrogen atoms does it contain? Draw the line bond structures of following types of hydrocarbons using four carbon atoms: 2. a. two noncyclic alkanes b. two cycloalkanes Give the molecular formula, the line bond structural formula, the condensed structural formula, and the skeletal formula for pentane 3. Molecular formula: Line bond structural formula: Condensed structural formula: Skeletal formulaarrow_forward
- Based on the molecular formula, determine whether each compound is an alkane, alkene, or alkyne. (Assume that the hydrocarbons are noncyclic and there is no more than one multiple bond.) a. C,H12 b. CH, c. C;H12 d. CH22arrow_forwardA saturated hydrocarbon Y with molecular formula of C4H8 exists as a pair of cis andtrans isomers:a. a. Draw the structural formulae for both cis and trans isomersb. b. Explain why Y exhibits geometrical isomersarrow_forwardI. Classify the following hydrocarbons as alkane, alkene, alkyne, or aromatic hydrocarbon. 1. Benzene 2. Pentene 3. Ethane 4. Hexyne 5. Butenearrow_forward
- Naming Organic Structures: 1. Give the IUPAC name for each of the following: CH2-CH3 H3C-CH2-Ç CH2 CH3 ČH CH3 a. CI. b.arrow_forwardDraw a condensed formula for each of the following compounds:a. trans-2, 2-dimethyl-2-hexeneb. 3-methyl-1, 5-nonadienec. cis-1, 2-dibromo-1-pentened. unsym. tert. pentyl n-propylethylenearrow_forwardQ1. Name each of the following structures by the IUPAC system? B. W C. Q2. Write the structural formula for the following? A. 1,4-dichloro-2-pentene B. 2-bromo-1,3-pentadiene C. vinyl cyclohexane Q3. Which of the following compounds can exist as cis- trans isomers? Draw their structures? A.. pentene B. 2-methyl-2-pentene Q3. Writ an equitation for the A. Reaction for bromine at room temperature 1. Propene B. The acid catalyzed addition of water to 1. 3-hexene Q4. For the addition of HBr to 3-methyl-1-butene, two products were observed. Show the reaction mechanism to explain the formation of both products. HBr - 3-methyl-1-butene major minorarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning  World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License