
Interpretation:
The structure of the compound has to be drawn, and Z or E stereochemistry has to be assigned to the product which is underwent E2 reaction with NaOH.
Concept introduction:
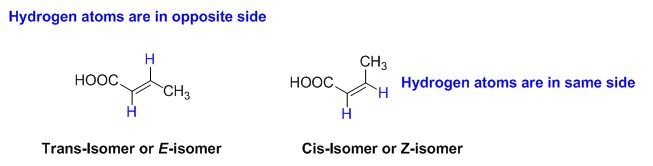
Z or E stereochemistry:
The two similar groups (or higher priority groups) are in same side in double bond of
Example:
E2 elimination:
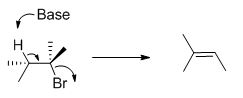
Given information:
The molecule is given in Ball-and-stick model and it undergoes E2 elimination with NaOH.
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- 1.) Write the products of this reaction. Show stereochemistry, and if possible, assign R or S.arrow_forwardThe reaction of tert-butyl cyclopentyl ether with trifluoroacetic acid is shown below. Explain what kind of reaction this is (SN1, SN2, E1, E2...) and show the mechanism for this transformation.arrow_forwardGive the major and minor products of the following E1 reaction and identify each and name each productarrow_forward
- Which of the statements below are correct about characteristics of an E2 reaction? i) Forms alkene product ii) Requires strong base iii) Minimum of one step iv) Concerted mechanismarrow_forwardWhich of the following hexachlorocyclohexanes is the least reactive in an E2 reaction?arrow_forwardWhen treated with NaOH, the bromide below gives an alkene by the E2 mechanism, by elimination of the H atom indicated by the arrow: (a) Draw the Newman projection from which elimination takes place. (b) Draw the mechanism. (c) Draw the product with the proper stereochemistry. (d) Assign the proper stereochemical descriptor to the product. (e) Give the rate equationarrow_forward
- Identify the reaction of the following reaction: formation of (R)-1-methoxypentane from (S)-1-bromopentane a. SN2 b. E2 c. SN1 d. E1arrow_forwardHow many distinct alkene products are possible when the alkyl iodide below undergoes E2 elimination?arrow_forwardClassify the carbocations given below as 1 °, 2 or 3 °, and write the product that will form the SN2 reaction on the most efficient substrate in a basic aqueous environment (H2O / HO).arrow_forward
- Which of the reactions below are correctly identified? These are SN2 reactions:arrow_forwardWhat product(s) would you expect to obtain from the following SN2 reaction?arrow_forwardAlthough there are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane, one stereoisomer reacts 7000 times more slowly than any of the others in an E2 elimination. Draw the structure of this isomer and explain why this is so.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

