
Concept explainers
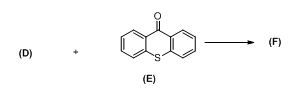
The antipsychotic drug flupentixol is prepared by the following scheme:
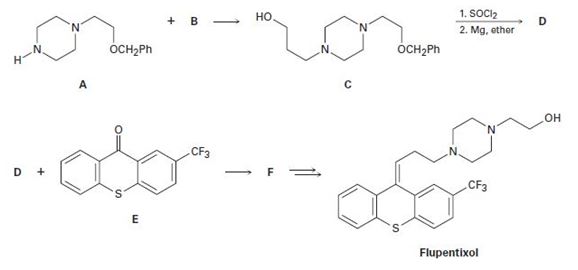
a)
The alkyl chloride, structure of D has to be identified and R and S configuration has to be assigned for the products.
Concept introduction:
R and S nomenclature: it is used to assign the molecule using CIP rules.
The CIP rules are as follows:
Select the chiral carbon and assign the numbers according to the decreasing atomic mass of atoms attached to it.
If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
Chiral: A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral: A molecule is superimposable on its mirror image is called achiral molecule.
Answer to Problem 81AP
The reaction is given below,
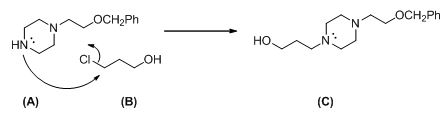
Explanation of Solution
Given information:
The given reactant shown below,
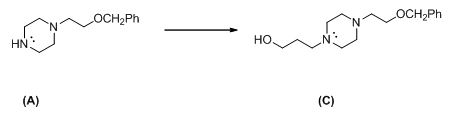
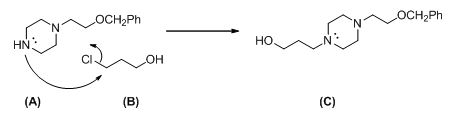
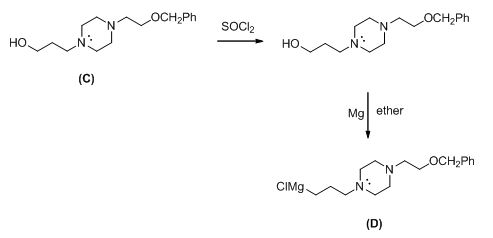
The given compound (A) reacts with chloro propanol gives the compound (C).
The reaction is given below,
The alkyl chloride, structure of D is given and R and S configuration is assigned for the products.
b)
Interpretation:
The alkyl chloride, structure of D has to be identified and R and S configuration has to be assigned for the products.
Concept introduction:
R and S nomenclature: it is used to assign the molecule using CIP rules.
The CIP rules are as follows:
Select the chiral carbon and assign the numbers according to the decreasing atomic mass of atoms attached to it.
If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
Chiral: A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral: A molecule is superimposable on its mirror image is called achiral molecule.
Answer to Problem 81AP
The Grignard reagent is given below,
Explanation of Solution
Given information:
The given reactant shown below,
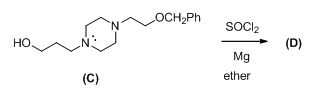
The Grignard reagent is given below,
The compound (c) reaction with thionyl chloride gives chlorinated compound, then it is reacting with magnesium gives the Grignard reagent (D).
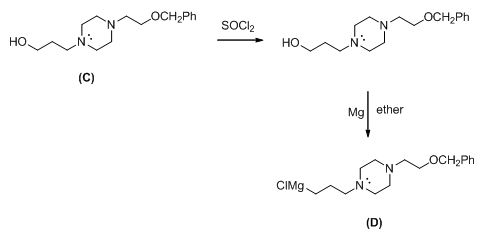
The alkyl chloride, structure of D is given and R and S configuration is assigned for the products.
c)
Interpretation
The alkyl chloride, structure of D has to be identified and R and S configuration has to be assigned for the products.
Concept introduction:
R and S nomenclature: it is used to assign the molecule using CIP rules.
The CIP rules are as follows:
Select the chiral carbon and assign the numbers according to the decreasing atomic mass of atoms attached to it.
If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
Chiral: A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral: A molecule is superimposable on its mirror image is called achiral molecule.
Answer to Problem 81AP
The pair of enantiomers is given below,
Explanation of Solution
Given information:
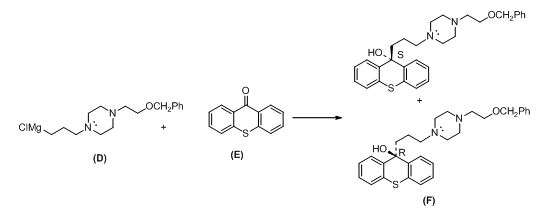
The given reactant shown below,
The pair of enantiomers is given below,
The compound (D) reaction with compound (E) gives enantiomer of corresponding products.
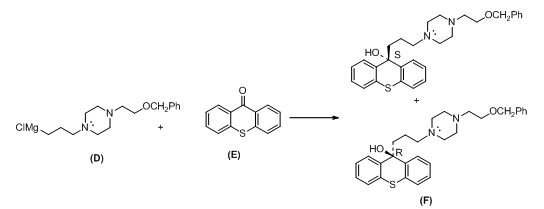
The alkyl chloride, structure of D is given and R and S configuration is assigned for the products.
d)
Interpretation:
The alkyl chloride, structure of D has to be identified and R and S configuration has to be assigned for the products.
Concept introduction:
R and S nomenclature: it is used to assign the molecule using CIP rules.
The CIP rules are as follows:
Select the chiral carbon and assign the numbers according to the decreasing atomic mass of atoms attached to it.
If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
Chiral: A molecule is non superimposable on its mirror image is called chiral molecule.
Four different atoms attached to a carbon atom is called chiral molecule.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Enantiomers: A compound which is non-superimposable mirror image is called enantiomers.
Diastereomers: A compound which is non-superimposable and non-mirror image is called diastereomers.
Racemic mixture: A racemic mixture is simply a mixture containing an equal amount of each enantiomer.
Achiral: A molecule is superimposable on its mirror image is called achiral molecule.
Answer to Problem 81AP
The compound (F) undergoes hydrolysis reaction gives E and Z isomers of corresponding products.
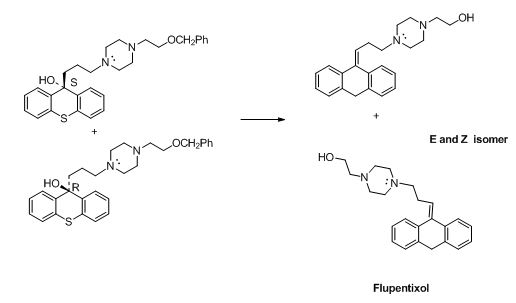
Explanation of Solution
Given information:
The given reactant shown below,
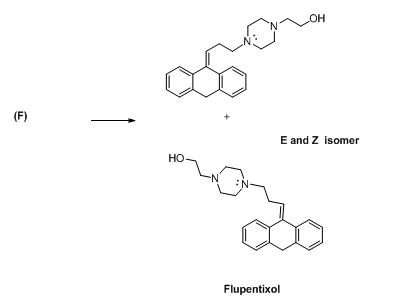
The compound (F) undergoes hydrolysis reaction gives E and Z isomers of corresponding products.
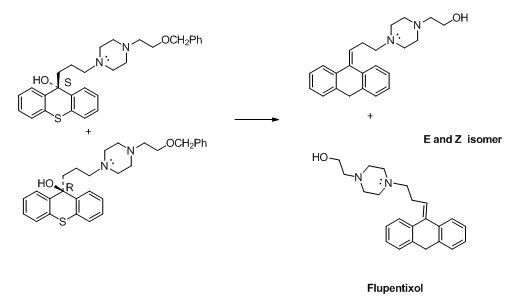
The alkyl chloride, structure of D is given and R and S configuration is assigned for the products.
Want to see more full solutions like this?
Chapter 11 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- Identify the pericyclic reaction benzocyclobutane E undergoes on heating, give astructure for intermediate H, and provide a mechanism that accounts for itsformation.arrow_forwardSuggest mechanisms for the following reactionsarrow_forwardPropose a synthesis of the anti-inflammatory drug Ibuprofen from benzene. Show all reagents and all intermediate structures. Assume that ortho and para isomers can be separatedarrow_forward
- Show how the following starting materials are converted to the givenproduct by a series of two pericyclic reactions. Account for the observedstereochemistry.arrow_forwardDescribe how the following compounds can be prepared from compoundscontaining no more than six carbons. (You can also use triphenylphosphine.)arrow_forwardSynthesize the following compound from benzonitrile (C6H5CN):arrow_forward
- Compound A reacts with 1 molar equivalent(s) of hydrogen upon catalytic hydrogenation. A undergoes reaction with ozone, followed by Zn treatment, to give: Propose a structure for A.arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forward
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.arrow_forwardFollowing is the structural formula of Surfynol, a defoaming surfactant. Describe the synthesis of this compound from acetylene and a ketone. How many stereoisomers are possible for Surfynol?arrow_forwardThe sex attractant of the housefly has the formula C23 H46. When treated with warm potassium permanganate, this phero- mone gives two products: CH3(CH2)12COOH and CH3(CH2)7COOH. Suggest a structure for this sex attractant. Explain which part of the structure is uncertain.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

