
Interpretation:
The faster reactant is to be identified from SN2 reaction with OH-.
Concept introduction:
SN2 reaction:
Tosylated compound is reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
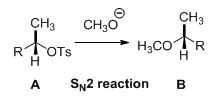
Given information:
a.
The given compound is shown below,
CH3I and CH3Br
b.
The given compound is shown below,
CH3CH2I in ethanol and CH3CH2I in dim ethylsulf oxide (DMSO)
c.
The given compound is shown below,
(CH3)3CCl and CH3Cl
d.
The given compound is shown below,
CH2 = CH2 Br or CH2 = CHCH2Br
Trending nowThis is a popular solution!

Chapter 11 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- Rank the following compounds in order of their reaction rates in an SN1 reaction with HOCH3 with 1 being the fastest reaction and 4 being the slowestarrow_forwardrank these from least to most reactive in nucleophilic acyl substitution with a nucleophile I)CH3COOC2H5 II) CH3COO-Na+ III)CH3COCl IV) CH3CONH2arrow_forwardQ2: Give the structure of the possible Claisen condensation product from the following reaction .Tell which , If any you would expect to predominate in each case .(a) CH3CO2Et + CH3CH2CO2Et (b) C6H6CO2Et + C6H5CH2CO2Et(c) EtOCO2Et + Cyclohexanones (d) C6H5CHO + CH3CO2Etarrow_forward
- The following questions ask you to choose which of the pair reactions shown has been correctly identified as faster. Choose all correct answers with explanation In the SN2 displacement by iodide of CH3Cl or of CH3OTs, CH3OTs reacts faster. In the SN2 displacement by acetate of bromoethane or bromocyclohexane, bromocyclohexane reacts faster. In the SN2 displacement by CH3CH2O- or by CN- on 2-bromopropane, CN- reacts faster. n the SN2 displacement by HC≡C- on bromomethane in benzene or in acetonitrile, reaction in benzene goes faster.arrow_forwardExplain how the reaction of (CH3)2CHCH(Cl)CH3 with H2O yields two substitutionproducts, (CH3)2CHCH(OH)CH3 and (CH3)2C(OH)CH2CH3arrow_forwardDraw the four resonance structures formed during bromination of methoxybenzene, CH3OC6H5, with Br2/FeBr3 to give para-bromomethoxybenzene and then circle the most stable resonance form.arrow_forward
- When allyl bromide is refluxed with magnesium metal in ether solvent, the product formed is 1,5-hexadiene. (C6H10). What is the curved arrow mechanism for this reaction?arrow_forwardDraw the favored product(s) of the following reactions (organic chemistry)arrow_forwardDraw a structural formula for the major organic product of each reaction and specify the most likely mechanism by which each is formed. (g) CH3CH2ONa++CH2=CHCH2Clethanolarrow_forward
- When 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observedarrow_forwardWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observed.(c) Use a Newman projection of the transition state to predict the major product of elimination of (2S,3R)-2-bromo-3-phenylbutanearrow_forwardProvide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d belowarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
