
Concept explainers
(a)
Interpretation:
The given statement is true or false should be determined.
The IUPAC name of an alkene is derived from the name of the longest carbon chain that contains the carbon-carbon double bond.
Concept Introduction:
The IUPAC name is the set of rules that is created and applied by the International Union of pure and Applied Chemistry to generate systematic proper name of chemical compounds. This nomenclature is used to describe and identify the type and position of functional groups, side chains, double or triple bonds etc. in alphabetical order to provide systematic name to compound.
Cis-trans stereoisomerisms are seen in alkenes compounds. Due to presence of two different groups bonded to carbon of carbon-carbon double bond, restricted action occurs in it and results into cis-trans isomerism. If same groups are present on the same side, then the isomer is called as cis-isomer and the same groups are present on different side the isomer is called as trans-isomer.
Answer to Problem 11P
The given statement is true.
Explanation of Solution
As per general rules of
(b)
Interpretation:
The given statement is true or false should be determined.
The IUPAC name of
Concept Introduction:
The IUPAC name is the set of rules that is created and applied by the International Union of pure and Applied Chemistry to generate systematic proper name of chemical compounds. This nomenclature is used to describe and identify the type and position of functional groups, side chains, double or triple bonds etc. in alphabetical order to provide systematic name to compound.
Alkenes are the compounds with very weak London dispersion forces and are nonpolar. They contain same skeleton structure and physical properties like alkanes but liquid at room temperature.
Cis-trans stereoisomerisms are seen in alkenes compounds. Due to presence of two different groups bonded to carbon of carbon-carbon double bond, restricted action occurs in it and results into cis-trans isomerism.
If same groups are present on the same side, then the isomer is called as cis- isomer and the same groups are present on different side the isomer is called as trans- isomer.
Answer to Problem 11P
The given statement is false.
Explanation of Solution
IUPAC name of
Therefore, the given statement is false.
(c)
Interpretation:
The given statement is true or false should be determined.
2-Methyl-2-butene shows cis-trans isomerism.
Concept Introduction:
The IUPAC name is the set of rules that is created and applied by the International Union of pure and Applied Chemistry to generate systematic proper name of chemical compounds. This nomenclature is used to describe and identify the type and position of functional groups, side chains, double or triple bonds etc. in alphabetical order to provide systematic name to compound.
Alkenes are the compounds with very weak London dispersion forces and are nonpolar. They contain same skeleton structure and physical properties like alkanes but liquid at room temperature.
Cis-trans stereoisomerisms are seen in alkenes compounds. Due to presence of two different groups bonded to carbon of carbon-carbon double bond, restricted action occurs in it and results into cis-trans isomerism.
If same groups are present on the same side, then the isomer is called as cis- isomer and the same groups are present on different side the isomer is called as trans- isomer.
Answer to Problem 11P
The given statement is false.
Explanation of Solution
2-Methyl-2-butene contains two methyl groups present on carbon atom of carbon-carbon double bond other carbon contains also methyl group.
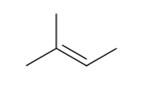
To get cis and trans isomers, two different groups should be present on the carbon atoms of the carbons with double bond.
Thus, the statement is false.
(d)
Interpretation:
The given statement is true or false should be determined.
1,2-dimethylcyclohexene shows cis-trans isomerism.
Concept Introduction:
The IUPAC name is the set of rules that is created and applied by the International Union of pure and Applied Chemistry to generate systematic proper name of chemical compounds. This nomenclature is used to describe and identify the type and position of functional groups, side chains, double or triple bonds etc. in alphabetical order to provide systematic name to compound.
Alkenes are the compounds with very weak London dispersion forces and are nonpolar. They contain same skeleton structure and physical properties like alkanes but liquid at room temperature.
Cis-trans stereoisomerisms are seen in alkenes compounds. Due to presence of two different groups bonded to carbon of carbon-carbon double bond, restricted action occurs in it and results into cis-trans isomerism.
If same groups are present on the same side then the isomer is called as cis- isomer and the same groups are present on different side the isomer is called as trans- isomer.
Answer to Problem 11P
The given statement is false.
Explanation of Solution
Following are the structures of the isomers of 1,2-dimethylcyclohexane. Form this, we can see that all four atoms of carbon-carbon double bond are lying in same plane.
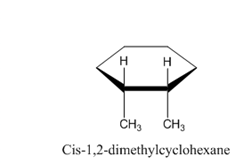
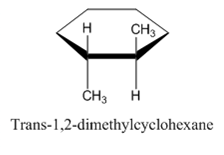
Therefore, the given statement is false.
(e)
Interpretation:
The given statement is true or false should be determined.
The IUPAC name of
Concept Introduction:
The IUPAC name is the set of rules that is created and applied by the International Union of pure and Applied Chemistry to generate systematic proper name of chemical compounds. This nomenclature is used to describe and identify the type and position of functional groups, side chains, double or triple bonds etc. in alphabetical order to provide systematic name to compound.
Alkenes are the compounds with very weak London dispersion forces and are nonpolar. They contain same skeleton structure and physical properties like alkanes but liquid at room temperature.
Cis-trans stereoisomerisms are seen in alkenes compounds. Due to presence of two different groups bonded to carbon of carbon-carbon double bond, restricted action occurs in it and results into cis-trans isomerism.
If same groups are present on the same side then the isomer is called as cis- isomer and the same groups are present on different side the isomer is called as trans- isomer.
Answer to Problem 11P
The given statement is true.
Explanation of Solution
The IUPAC name of
(f)
Interpretation:
The given statement is true or false should be determined.
1,3-Butadiene has two carbon-carbon double bonds and 22 =4 stereoisomers are possible for it.
Concept Introduction:
The IUPAC name is the set of rules that is created and applied by the International Union of pure and Applied Chemistry to generate systematic proper name of chemical compounds. This nomenclature is used to describe and identify the type and position of functional groups, side chains, double or triple bonds etc. in alphabetical order to provide systematic name to compound.
Alkenes are the compounds with very weak London dispersion forces and are nonpolar. They contain same skeleton structure and physical properties like alkanes but liquid at room temperature.
Cis-trans stereoisomerisms are seen in alkenes compounds. Due to presence of two different groups bonded to carbon of carbon-carbon double bond, restricted action occurs in it and results into cis-trans isomerism.
If same groups are present on the same side, then the isomer is called as cis- isomer and the same groups are present on different side the isomer is called as trans- isomer.
Answer to Problem 11P
The given statement is false.
Explanation of Solution
Following is the structure of 1,3-Butadiene. There are no stereocenters are present in the structure of 1,3-Butadiene. There should be presence of different groups for isomerism.
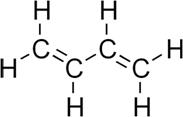
Therefore, the given statement is false.
Want to see more full solutions like this?
Chapter 12 Solutions
INTRO.TO GENERAL,ORGANIC+BIOCHEM.(LL)
- Please do the following question. Step by step A. 1-butyne + HgSO4, H2SO4 and H2O B. 2-methyl-1,3-butadiene + HC ≡ C – C ≡ Narrow_forwardhow to name cyclic or acyclic alkanes ? what are the steps and rules that I have to follow to name anyone that comes my wayarrow_forward1. What is the correct IUPAC name for 2-propylpentane (For Alkene and Alkyne Naming, follow this format “Branch-Carbon Prefix-Functional Group Suffix”) e.g. 3-methylhex-2-ene 2. What is the correct IUPAC name for 3-butyl-2-isopropylpentane 3. What is the correct IUPAC name for 2,5-Dipropylhexane 4. What is the correct IUPAC name for 5-butyl-6-ethyl-4-methylhex-1-ene 5. What is the correct IUPAC name for 3,4-dimethylhex-5-ene-1-ynearrow_forward
- What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2?arrow_forward13-3 What is the difference in structure between a saturated and an unsaturated hydrocarbon?arrow_forwardI am confused as to which compound from the following: cyclhexane, ethyne, hept-2-ene, Ch2(CH2)14Ch2OH, dode-1-yne, Ch3Ch2CO2H,Propan-2-ol, Ch3(Ch2)14CO2H; is miscible in water, is an insoluble gas, is a water insosuble solid, is a wter insoluble liquid, is a water insoluble liquid- it burns clean, is a water insuloble liquid-it burns dirty, us water mscible. What do I look for to assign each compound to its coorect characterisitcs.arrow_forward
- Kindly answer the following questions from 1 to 3. As per your guidelines, only solve the first three questions. 1. Draw the Lewis structures and predict the hybridization (sp, sp2, sp3), geometry (linear, trigonal, tetrahedral) and bond angles (180°, 120°, 109.5°) for the central atoms in the following compounds. 2. Give the systematic (IUPAC) name for each of the following alkanes, cycloalkanes, bicycloalkanes and alkyl halides. 3. Given the IUPAC name, draw the structure of the following compounds.arrow_forwardWrite IUPAC name of the following alkanes ? e ) ,f)arrow_forwardConvert 2- butyne to trans – 2- butane.arrow_forward
- So, this is one of the questions on our quizzes. As far as I know, it's supposed to be 2-diethyl-5-methylhexane but it's not in the choices. Correct me if I am wrong. The quiz is about IUPAC naming of alkanes.arrow_forwardDetermine the Anti-Markovnikov Product prop-1-ene + H2O—> _________ (For Alkene and Alkyne Naming, follow this format “Branch-Carbon Prefix-Functional Group Suffix”) e.g. hex-2-enearrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

