
(a)
To determine: The characteristic infrared absorptions of the
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or
(a)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
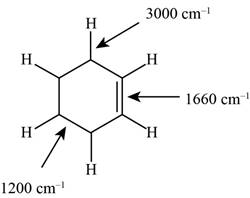
Figure 1
Explanation of Solution
The structure of the given molecule is,
Figure 2
The possibilities of the IR stretching frequencies for the given molecule are shown as,
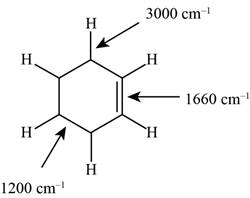
Figure 1
So, the stretching frequencies of
(b)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(b)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
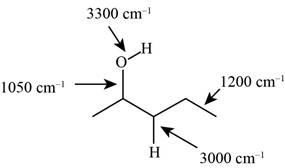
Figure 3
Explanation of Solution
The structure of the given molecule is,
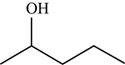
Figure 4
The possibilities of the IR stretching frequencies for the given molecule are shown as,
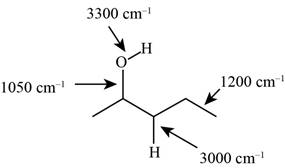
Figure 3
So, the stretching frequencies of
(c)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(c)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
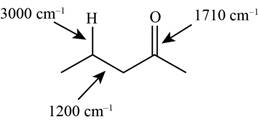
Figure 5
Explanation of Solution
The structure of the given molecule is,
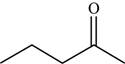
Figure 6
The possibilities of the IR stretching frequencies for the given molecule are shown as,
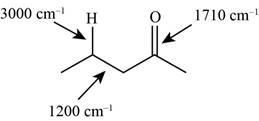
Figure 5
So, the stretching frequencies of
(d)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(d)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
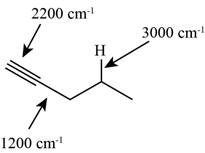
Figure 7
Explanation of Solution
The structure of the given molecule is,
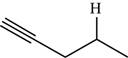
Figure 8
The possibilities of the IR stretching frequencies for the given molecule are shown as,

Figure 7
So, the stretching frequencies of
(e)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(e)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
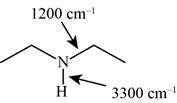
Figure 9
Explanation of Solution
The structure of the given molecule is,
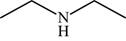
Figure 10
The possibilities of the IR stretching frequencies for the given molecule are shown as,
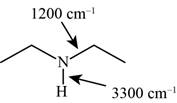
Figure 9
So, the stretching frequencies of
(f)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule is to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(f)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
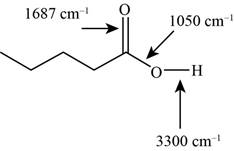
Figure 11
Explanation of Solution
The structure of the given molecule is,
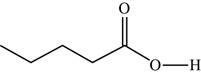
Figure 12
The possibilities of the IR stretching frequencies for the given molecule are shown as,

Figure 11
So, the stretching frequencies of
(g)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(g)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
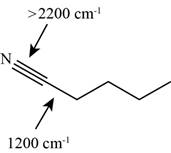
Figure 13
Explanation of Solution
The structure of the given molecule is,
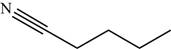
Figure 14
The possibilities of the IR stretching frequencies for the given molecule are shown as,
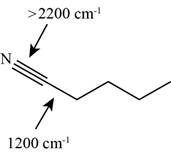
Figure 13
So, the stretching frequencies of
(h)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(h)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
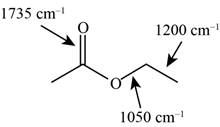
Figure 15
Explanation of Solution
The structure of the given molecule is,
Figure 16
The possibilities of the IR stretching frequencies for the given molecule are shown as,
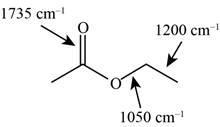
Figure 15
So, the stretching frequencies of
(i)
To determine: The characteristic infrared absorptions of the functional groups in the given molecule.
Interpretation: The characteristic infrared absorptions of the functional groups in the given molecule are to be predicted.
Concept introduction: An IR spectrum is a graph for the energy absorbed by a molecule as a function of the frequency or wavelength of light. Alkanes, alkenes and alkynes have characteristic
(i)
Answer to Problem 12.12SP
The characteristic infrared absorptions of the functional groups in the given molecule are,
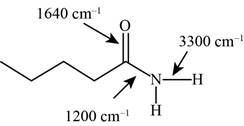
Figure 17
Explanation of Solution
The structure of the given molecule is,
Figure 18
The possibilities of the IR stretching frequencies for the given molecule are shown as,

Figure 17
So, the stretching frequencies of
Want to see more full solutions like this?
Chapter 12 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
- Which of the following is most UNLIKELY to absorb in the UV-Vis region? a. chlorophyll b. heptane c. nitrophenol d. beta-carotenearrow_forwardMatch set A (common active ingredients of insect repellants) to set B (known properties). Then, match set B to set C (structure). Then, match set C to set A. Set A Set B 1. Citriodiol a. Considered as biopesticide b. Lemon eucalyptus oil c. Gold standard repellant 2. Picaridin 3. DEET 4. Citronellol and Geraniol 5. IR-3535 d. Citronella oil e. Repellant of choice for malarial prevention Set C CH3 а. d. CH3 HO OH H3C CH3 H3C CH3 b. е. с. OH OHarrow_forwardFor each organic compound in the table below, enter the locant of the highlighted side chain. CH3 CH₂ CH₂ H CH3 · C CH₂- CH CH3 CH₂ CH3- compound CH₂ I CH₂- CH CH | CH₂ CH3- — - CH₂ | CH₂-C. CH3 | — ぎーぎ CH3 locant of highlighted side chain 0 0arrow_forward
- Data for Problem 2 ZOO 180 150 120 140 100 80 770 дом -8 60 40 20 10 9 a 5 015 100 80 Relative Intensity 8 ส 20 20 40 50 ppm septet 3 2 60 70 80 90 100 m/zarrow_forwardWhich of the following compounds has a UV spectroscopic signal at the lowest absorption energy? II IV A. I B. II C. II D. IVarrow_forward1. Which of the following compounds is separable by a physical method such as distillation? D A B.arrow_forward
- Please provide both answers.arrow_forwardThe structure of the molecule cyclohexene is Does the absorption of ultraviolet light by cyclohexene occur at longer or at shorter wavelengths than the absorption by benzene? Explain.arrow_forward1.When zg of a certain hydrocarbon was burnt completely 0.62g of co2 and 0.25g of H2O Was obtained te emperical formular of the compound is A.CHZO B.CHZ C. CH D.C2H2 2.The atomic number of iron is 24 what is the correct electronic configuration of FE3+ 3.The shape of PCLS is 4.The function of electron gun in the mass spectrometer is A.Fires fast moving electrons B.lonize gasoeus atoms producing +ve ions C.Accelerates the ionize gasoeus atoms D.Vapourise the element 5.The coordination number and oxidation state of element E the complex K3(E(CN6) A.4 and +2 B.4 and +3 C.6 and +2 D.6 and +3 6.A substance that gives a green flame on tes coulour on heating produced an acidic gas and another gas that rekindled a glowing splint which of the following could it be A.Barium nitrate B. Barium carbonate C. Copper caronate D.Barium nitratearrow_forward
- Molecular Structure and Properties 1. Identify the functional groups in the following molecules. Tell how you could differentiate the following pairs of compounds with the help of ONLY infrared spectroscopy. (see Table at pp.6,7) a) ´d d CH3 b) c) d) CH3 CH3 om & CH3 OH OH OHarrow_forwardFor each of the five molecules shown below, tell whether it will be expected to show ultraviolet absorptions in the 200 to 400 nm range or not. Explain your answer for each. CH3 b. C. Indole OH Aspirin d.arrow_forwardMost alkenes absorb at a shorter wavelength than alkadienes. Explain.arrow_forward

 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

