
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.9A, Problem 12.4P
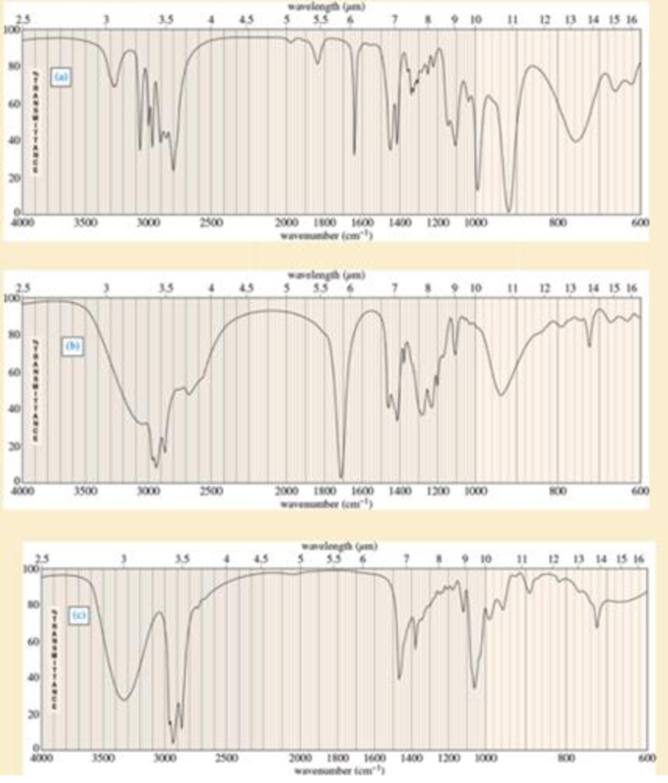
Spectra are given for three compounds. Each compound has one or more of the following
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The infrared spectrum of a compound is unique to the types and number of bonds in the molecule. Draw the structures of salicylic acid and aspirin. What are the key differences of the IR spectrum of aspirin with that of the starting material, salicylic acid? What are the similarities? Interpret the IR spectra of each by assigning the major peaks.
For each organic compound in the table below, enter the locant of the highlighted side chain.
CH3
CH₂
CH₂
H
CH3 · C CH₂- CH CH3
CH₂
CH3-
compound
CH₂
I
CH₂- CH CH
|
CH₂
CH3-
—
-
CH₂
|
CH₂-C. CH3
|
—
ぎーぎ
CH3
locant of highlighted side
chain
0
0
In the compound below, identify the functional group in the box.
A Functional Group in a Compound
A
hydroxyl
B ketone
с aldehyde
D amine
Br
O
||
C
H
Chapter 12 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 12.3 - Complete the following conversion table. (cm1)...Ch. 12.5 - Which of the bonds shown in red are expected to...Ch. 12.7C - For each hydrocarbon spectrum, determine whether...Ch. 12.9A - Spectra are given for three compounds. Each...Ch. 12.10 - The infrared spectra for three compounds are...Ch. 12.12 - Prob. 12.6PCh. 12.14B - Identify which of these four mass spectra indicate...Ch. 12.15A - Show the fragmentation that accounts for the...Ch. 12.15A - Show the fragmentations that give rise to the...Ch. 12.15B - Ethers are not easily differentiated by their...
Ch. 12.15C - Prob. 12.11PCh. 12 - Prob. 12.12SPCh. 12 - Prob. 12.13SPCh. 12 - All of the following compounds absorb infrared...Ch. 12 - Prob. 12.15SPCh. 12 - Four infrared spectra are shown, corresponding to...Ch. 12 - Predict the masses and the structures of the most...Ch. 12 - Prob. 12.18SPCh. 12 - Prob. 12.19SPCh. 12 - (A true story) While organizing the undergraduate...Ch. 12 - Prob. 12.21SPCh. 12 - Prob. 12.22SPCh. 12 - An unknown, foul-smelling hydrocarbon gives the...Ch. 12 - covered a synthesis of alkynes by a double...Ch. 12 - Three IR spectra are shown, corresponding to three...Ch. 12 - Prob. 12.26SPCh. 12 - Prob. 12.27SPCh. 12 - Prob. 12.28SPCh. 12 - The ultimate test of fluency in MS and IR is...Ch. 12 - Prob. 12.30SPCh. 12 - Consider the following four structures, followed...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Here is the chemical structure of 2-bromobutane: Н Η Н Н HHHH C C- C C-H H :Br Η Н .. Decide whether each molecule in the table below is another molecule of 2-bromobutane, a molecule of an isomer of 2-bromobutane, or a molecule of an entirely different compound. molecule CH₂ CH₂-CH₂-CH-Br CH₂ CH₂ -CH₂ Br HI H H H Η H_ H-C- C-C- H-C-H H H relationship to 2-bromobutane (Choose one) (Choose one) Br: a molecule of an isomer of 2-bromobutane ▼arrow_forwardWhat are these 2?arrow_forwardWrite the systematic name of each organic molecule: (10 HO—C—CH–CH2–CH–CH2–CH2−C CH3 I CH3 structure || HO—C—CH—CH,—OH CH3 CH3 CI-CH₂-CH-CH₂-C-OH name 0arrow_forward
- 2. The full structural formulae of three organic compounds, P, Q and R, are shown below. H H H H HH HHHH Н-С -с- Н H-C- C - C = C – H H - C - C = C – C – H H H H H Q P (a) State one similarity between P, Q and R in terms of their molecular formulae. (b) Name the homologous series that compounds P, Q and R belong to. (c) State one similarity between Q and R in terms of chemical bonding id) Which of these compounds are isomers? Explain your answer.arrow_forwardDetermine the compound (name or structure) from the data. Explain features from each data. Molecular formula: C6H5Br .arrow_forwardO—CH,CH,CH HBrarrow_forward
- Compound 1 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Compound 2 has molecular formula C7H15Br. It shows two signals in the 1H-NMR spectrum, one at 1.08 ppm and one at 1.59 ppm. The relative integrals of these two signals are 3 and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardWrite the systematic name of each organic molecule: CH3 structure -C−NH–CH2−CH2CH3 || H₂N-C-CH₂ -CH2–CH2–CH2–CH3 CH3 CH2–CH2–NH–C–CH2–CH3 name 11 0 0arrow_forwardMgBr Mg C,H,OC,H, MgBr o-CH O *MgBr C,H,OC,H, + CH,OH H,O, H,SO, O "MgBr OH Data Glassware oven-dried, assembled, placed under nitrogen (balloon). Materials: 10 mL anhydrous diethyl ether, 1.0 g Mg turnings. 4.5 mL bromobenzene. 10 mL more anhydrous diethyl ether. 2.5 ml methyl benzoate. Approximate initiation time: 15 min. Stir 10 min after addition of methyl benzoate. Transfer reaction mixture to 100 mL Erlenmeyer flask. Rinse reaction flask (2 x 20 mL diethyl ether). Acid quench solution: 25 mL 10 % aqueous sulfuric acid/15 g ice. Saturated NaCI solution: used 20 mL Hexanes: 20 mL Isolated: 1.82 g white solid What is the % yield of the reaction? What is the limiting reagent? Show complete calculations for the theoretical and % yield calculationsarrow_forward
- Given below are condensed structural formulas derived from the same molecular formula (C,H14). Identify which pair represents structural isomers, that is, different molecules. CH3 CH3 CH, CH3 CH, CH, CH-CH3 CH, CH-CH-CH3 CH3 CH, CH-CH,-CH2 CH3 CH, CH-CH CH, CH3 CH3 A and C O Cand D O Band D O D and Aarrow_forwardPlearrow_forwardCompound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY