
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.15A, Problem 12.9P
Show the fragmentations that give rise to the peaks at m/z 43, 57, and 85 in the mass spectrum of 2,4-dimethylpentane (Figure12-17).
| m/z | Abundance (% of base Peak) |
| 41 | 34 |
| 42 | 24 |
| 43(base peak) | 100 |
| 56 | 35 |
| 57 | 72 |
| 85 | 19 |
| 100 (M+) |
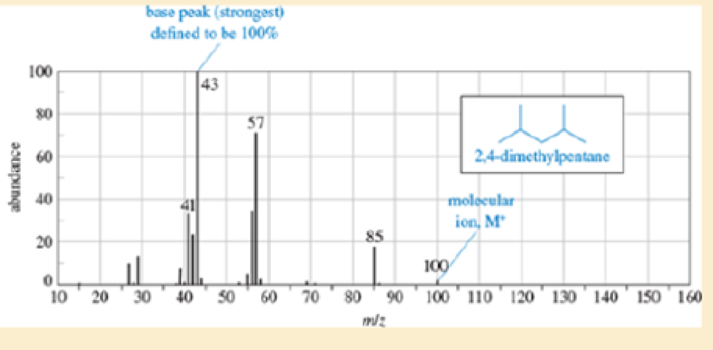
Figure 12-17
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The mass spectrum of 4-methoxybenzaldehyde B is shown in Figure 1. Assign
the labelled signals and state whether each of your assignments is a fragment
ion or a molecular ion.
4- methoxybenzaldehyde
MASS SPECTRUM
100
136
80
60
78
108
40
20
0.0+
0.0
40
80
120
160
m/z
Figure 1
Rel. Intensity
The following mass spectrum has a molecular ion and the M+2 ion. These molecular ions indicate the
presence of a specific atom in the molecule. Identify this atom.
Relative Intensity
100
80
8
40
20
0
ON
13C
Br
OCI
10
MS-NW-1039
20
30
40
50
60
m/z
70
80
90
100
What does the "(<1%)" in the mass spectrum mean?
Chapter 12 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 12.3 - Complete the following conversion table. (cm1)...Ch. 12.5 - Which of the bonds shown in red are expected to...Ch. 12.7C - For each hydrocarbon spectrum, determine whether...Ch. 12.9A - Spectra are given for three compounds. Each...Ch. 12.10 - The infrared spectra for three compounds are...Ch. 12.12 - Prob. 12.6PCh. 12.14B - Identify which of these four mass spectra indicate...Ch. 12.15A - Show the fragmentation that accounts for the...Ch. 12.15A - Show the fragmentations that give rise to the...Ch. 12.15B - Ethers are not easily differentiated by their...
Ch. 12.15C - Prob. 12.11PCh. 12 - Prob. 12.12SPCh. 12 - Prob. 12.13SPCh. 12 - All of the following compounds absorb infrared...Ch. 12 - Prob. 12.15SPCh. 12 - Four infrared spectra are shown, corresponding to...Ch. 12 - Predict the masses and the structures of the most...Ch. 12 - Prob. 12.18SPCh. 12 - Prob. 12.19SPCh. 12 - (A true story) While organizing the undergraduate...Ch. 12 - Prob. 12.21SPCh. 12 - Prob. 12.22SPCh. 12 - An unknown, foul-smelling hydrocarbon gives the...Ch. 12 - covered a synthesis of alkynes by a double...Ch. 12 - Three IR spectra are shown, corresponding to three...Ch. 12 - Prob. 12.26SPCh. 12 - Prob. 12.27SPCh. 12 - Prob. 12.28SPCh. 12 - The ultimate test of fluency in MS and IR is...Ch. 12 - Prob. 12.30SPCh. 12 - Consider the following four structures, followed...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. The mass spectrum of 3-hexanol is given below. 100 Intensity 55 80- 60- 40 40 20 20 59 73 3-Hexanol El MW = 102 OH 0 0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 150 m/z (a) Rationalize the small size of the molecular ion peak at m/z 102. (b) Assign structures to all the labeled species and rationalize their formation.arrow_forwardplease answer all the questions, 16-21. thank you.arrow_forward12. The mass spectrum of 2-methylhexane is shown below. What is the m/z value of the M* peak and of the base peak? Give possible structures of the fragments giving rise to the large peaks at m/z = 85,57, and 43. 100 - 80 - 40 20 50 60 70 80 90 100 10 20 30 40 m/z Relative Intensityarrow_forward
- Which of the following structures corresponds to the mass spectrum shown below? Relative Intensity 100 80- 60 40 20 MS-NW-0358 0-mt 20 40 CI 60 80 100 120 m/z 140 160 180 M+ = 216 200 220arrow_forwardCan I please have help with this?arrow_forwardLoss of which fragment from the parent molecule could account for the peak at 57 in the mass spectrum of 2-methylbutane? 100 -43 57 80 29 100 110 Relative Intensity 60 20 0 T T T T T T T 10 ZD 30 40 50 60 72 70 m/z 80 90 120arrow_forward
- The intensity of M+, M+1, and M+2 peak are 100.0, 6.8, 31.9. How many carbons are in the compound? (whole number digit only) 100 MS-NW-5495 80 60 40 10 20 30 40 50 60 70 80 90 100 110 m/z Relative Intensity 20arrow_forwardA low-intensity peak at ?/? = 31 can be seen in the mass spectrum of methyl acetate in Figure 3. Propose a fragment that could originate from methyl acetate and be responsible for this peak.arrow_forwardAn unknown molecule was run through WT's mass spectrometer and the following spectra was produce. What could the unknown compound be? MASS SPECTRUM 100 50 80- 43 60 98 40 27 20- 83 0.0 0.0 20 40 60 80 100 m/z Rel. Intensityarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY