
Concept explainers
(a)
Interpretation:
The model structure has to be converted into line drawing along with identification of Functional Group.
Concept Introduction:
Line structure is a simple and quick way to represent organic molecules without showing carbons and hydrogens present. Drawing a molecule in this way is very simple:
It follows the following guide line:
- Each carbon- carbon bond is represented by a line.
- Anywhere a line ends or begins, as well as any vertex where two lines meet represents a carbon atom.
- Any atom other than another’s carbon or hydrogen attached to a carbon must be shown.
- Since a neutral carbon atom form four bonds, all bonds not shown for any carbon are understood to be the number of carbon – hydrogen bonds needed to have.
Functional group: Organic compounds can be classified into various families according to the functional groups they contain. A functional group is a part of a larger molecule and is composed of a group of atoms that has characteristic structure and chemical reactivity.

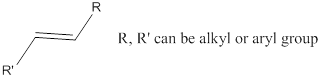
Ether: A class of organic compounds, where oxygen is attached to two alkyl or aryl groups.
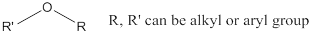
(b)
Interpretation:
The model structure has to be converted into line drawing along with identification of Functional Group.
Concept Introduction:
Line structure is a simple and quick way to represent organic molecules without showing carbons and hydrogens present. Drawing a molecule in this way is very simple:
It follows the following guide line:
- Each carbon- carbon bond is represented by a line.
- Anywhere a line ends or begins, as well as any vertex where two lines meet represents a carbon atom.
- Any atom other than another’s carbon or hydrogen attached to a carbon must be shown.
- Since a neutral carbon atom form four bonds, all bonds not shown for any carbon are understood to be the number of carbon – hydrogen bonds needed to have.
Functional group: Organic compounds can be classified into various families according to the functional groups they contain a functional group is a part of a larger molecule and is composed of a group of atoms that has characteristic structure and chemical reactivity.
Functional group:
An atom or group of atoms which has physical and chemical properties is known as functional group.
Ketone: A compound containing a carbonyl group

Alkene: Unsaturated hydrocarbons containing double bonds.
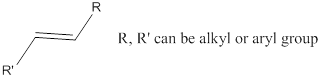
Ether: A class of organic compounds, where oxygen is attached to two alkyl or aryl groups.
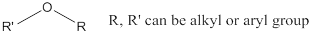
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Study Guide And Full Solutions Manual For Fundamentals Of General, Organic, And Biological Chemistry
- Give at least 10 examples of biological compounds having an alkyne functional group and identify the biochemical importance of each compound.arrow_forwardDraw Lewis structures for two compounds of formula C2H7N.arrow_forwardWith the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forward
- Give IUPAC names for the five isomers with the formula C6H14.arrow_forwardBalance the following equation, and tell how many moles of nickel will reactwith 9.81 mol of hydrochloric acid.arrow_forwardConsider the intermolecular forces present in a pure sample of each of the following compounds: CH₃CH₂OH and CH₃COCH₃. Identify the intermolecular forces that these compounds have in common.arrow_forward
- What is the empirical formula for C3H6O3? C3H6O3 C6H12O6 CH2O None of thesearrow_forwardConsider the following acids and their ionization constant, determine which conjugate base is HCOOH Ka = 1.7 x 10-4 (b) HCN Ka = 4.9 x 10-10arrow_forwardUsing the picture provided, match the correct name of each of the functional groups highlighted in blue.arrow_forward
- Convert the following structural formulas into condensed structures.arrow_forwardWrite the chemical formula for the conjugate base of each of the following acids: Carbonic Acid (H2CO3)___________________ Formic Acid (CH2O2)_____________________arrow_forwardIdentify the names of the following molecules.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





