
(a)
Interpretation:
The structural formula for the
Concept introduction:
The addition reactions are defined as the formation of one product by the combination of two or more products. For example, in halogenation, the addition of a halogen molecule results in the reduction of carbon-carbon double bond to carbon-carbon single bond and formation of
Answer to Problem 12.27E
The structural formula for the alkene with molecular formula
Explanation of Solution
The reaction is shown below.
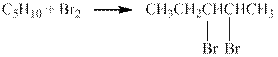
Figure 1
The product
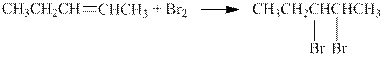
Figure 2
The structural formula for the alkene with molecular formula
(b)
Interpretation:
The structural formula for the alkene with molecular formula
Concept introduction:
The addition reactions are defined as the formation of one product by the combination of two or more products. For example, in halogenation, the addition of a halogen molecule results in the reduction of carbon-carbon double bond to carbon-carbon single bond and formation of haloalkane.
Answer to Problem 12.27E
The structural formula for the alkene with molecular formula
Explanation of Solution
The reaction is shown below.
The product in the given reaction is pentane which can be prepared by the hydrogenation on the
The structural formula for the alkene with molecular formula
(c)
Interpretation:
The structural formula for the alkene with molecular formula
Concept introduction:
The Markovnikov’s rule states that the addition of the
Answer to Problem 12.27E
The structural formula for the alkene with molecular formula
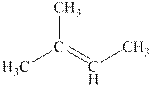
Explanation of Solution
The reaction is shown below.
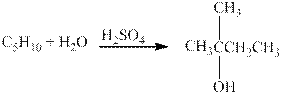
Figure 2
The product in the reaction is
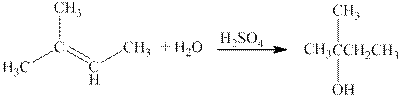
Figure 3
The structural formula for the alkene with molecular formula
(d)
Interpretation:
The structural formula for the alkene with molecular formula
Concept introduction:
The Markovnikov’s rule states that the addition of the
Answer to Problem 12.27E
The structural formula for the alkene with molecular formula
Explanation of Solution
The given reaction is shown below.

Figure 4
The product in the given reaction is
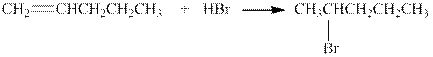
Figure 5
The structural formula for the alkene with molecular formula
Want to see more full solutions like this?
Chapter 12 Solutions
CHEMISTRY FOR TODAY+OWLV2 24 MO>IP<
- Define Oxidation of Alkanes ?arrow_forward1. What is the reaction for incomplete combustion of hexane? 2. Write pyrolysis of octane? 3. What is the reaction of the replacement of hydrogen atoms in propane by chlorine atoms in the presence of light?arrow_forwardExplain why alkenes are much more reactive than alkanes towards chlorine (CI2) or bromine (Br2) in the dark at room temperature, and why alkanes do not react with HCI (g) or HBr (g) whereas alkenes do.arrow_forward
- Draw structural formulas for these alkenes. Q.) 2-Chloropropenearrow_forwardExplain oxidation and reduction of alkynes ?arrow_forwardWrite the structure of the compound that will be produced in the following reaction? CH3 –C ≡ C–CH2– CH2 – CH3 + 2HBr→ a) Other than the combustion of alkanes, why are alkanes not reactive, or when they do react, they do so very slowly? B) What chemical reaction can alkenes and alkynes undergo that alkanes cannot? Explainarrow_forward
- Consider the reaction of gaseous propene with liquid water to produce liquid 2- propanol. a. Write the balanced chemical equation for this reaction including phase labels. b. What would the Law of Mass Action be? (The specific mathematical equation for K)arrow_forward1. Which of the following fuel has the lowest heat of combustion?a.Methaneb.Octanec.Coald.Ethanol 2. What is the process of breaking larger hydrocarbon molecules into smaller ones at low temperature through the use of catalyst in order to obtain higher quality of gasoline?a.Thermal crackingb.Catalytic crackingc.Catalytic reformingd.Catalytic combination 3. What is the most common oxidizing agent available in the atmosphere that can be used to generate fire?a.hydrogen peroxideb.oxygenc.ozoned.nitrous oxidearrow_forwardWhat type of product is formed in the reaction between an alkane and a halogen? A) alkene B) cycloalkane C) alkyl halide D) alkyne E) acid halidearrow_forward
- What mass of 2-bromopropane could be prepared from 25.5 g of propene? Assume a 100% yield of product.arrow_forwardConsider a sample of a hydrocarbon at 0.959 atm and 298 K. Upon combusting the entire sample in oxygen, you collect a mixture of gaseous carbon dioxide and water vapor at 1.51 atm and 375 K. This mixture has a density of 1.391 g/L and occupies a volume four times as large as that of the pure hydrocarbon. Determine the molecular formula of the hydrocarbon and name it.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





