
Concept explainers
(a)
Interpretation:
Predict the simple binary compound that can be formed by the use of electron configuration of the simple ions which are formed by those elements.
Concept Introduction:
Electron configuration describes the positions of electrons of an atom. Depending on that the periodic table is created. Periodic table further defines the simple ion that can be formed by any element. And using those ions, simple binary compounds can be produced.
Order in which orbitals fill to produce the atoms in periodic table as follows:
Simplest ion form by each group as follows:
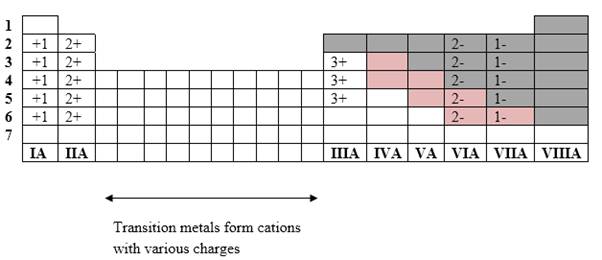
If it’s a compound made by a combination of metal and nonmetal elements, called as ionic compounds.
Ionic chemical compound must have a zero charge. If a compound contains ions;
1. Both positive ions (cations) and negative ions (anions) must be there.
2. No: of cations and anions collectively should produce a zero-net charge.
Answer to Problem 45CR
Explanation of Solution
Al and Cl
(b)
Interpretation:
Predict the simple binary compound that can be formed by the use of electron configuration of the simple ions which are formed by those elements.
Concept Introduction:
Electron configuration describes the positions of electrons of an atom. Depending on that the periodic table is created. Periodic table further defines the simple ion that can be formed by any element. And using those ions, simple binary compounds can be produced.
Order in which orbitals fill to produce the atoms in periodic table as follows:
Simplest ion form by each group as follows:
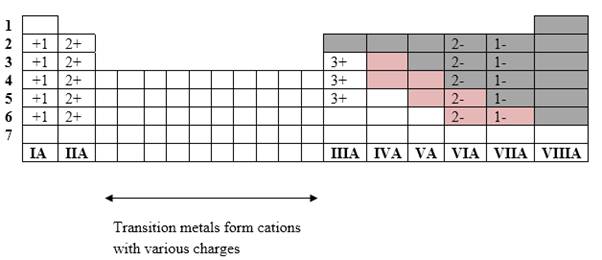
If it’s a compound made by a combination of metal and nonmetal elements, called as ionic compounds.
Ionic chemical compound must have a zero charge. If a compound contains ions;
1. Both positive ions (cations) and negative ions (anions) must be there.
2. No: of cations and anions collectively should produce a zero-net charge.
Answer to Problem 45CR
Explanation of Solution
Na and N
(c)
Interpretation:
Predict the simple binary compound that can be formed by the use of electron configuration of the simple ions which are formed by those elements.
Concept Introduction:
Electron configuration describes the positions of electrons of an atom. Depending on that the periodic table is created. Periodic table further defines the simple ion that can be formed by any element. And using those ions, simple binary compounds can be produced.
Order in which orbitals fill to produce the atoms in periodic table as follows:
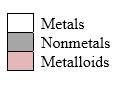
Simplest ion form by each group as follows:
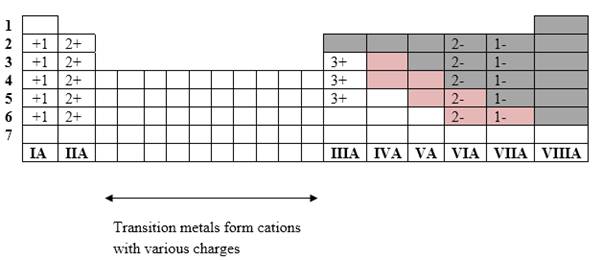
If it’s a compound made by a combination of metal and nonmetal elements, called as ionic compounds.
Ionic chemical compound must have a zero charge. If a compound contains ions;
1. Both positive ions (cations) and negative ions (anions) must be there.
2. No: of cations and anions collectively should produce a zero-net charge.
Answer to Problem 45CR
Explanation of Solution
Mg and S
(d)
Interpretation:
Predict the simple binary compound that can be formed by the use of electron configuration of the simple ions which are formed by those elements.
Concept Introduction:
Electron configuration describes the positions of electrons of an atom. Depending on that the periodic table is created. Periodic table further defines the simple ion that can be formed by any element. And using those ions, simple binary compounds can be produced.
Order in which orbitals fill to produce the atoms in periodic table as follows:
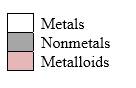
Simplest ion form by each group as follows:
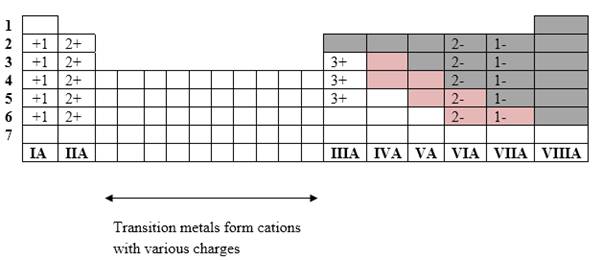
If it’s a compound made by a combination of metal and nonmetal elements, called as ionic compounds.
Ionic chemical compound must have a zero charge. If a compound contains ions;
1. Both positive ions (cations) and negative ions (anions) must be there.
2. No: of cations and anions collectively should produce a zero-net charge.
Answer to Problem 45CR
CaO.
Explanation of Solution
Ca and O
(e)
Interpretation:
Predict the simple binary compound that can be formed by the use of electron configuration of the simple ions which are formed by those elements.
Concept Introduction:
Electron configuration describes the positions of electrons of an atom. Depending on that the periodic table is created. Periodic table further defines the simple ion that can be formed by any element. And using those ions, simple binary compounds can be produced.
Order in which orbitals fill to produce the atoms in periodic table as follows:
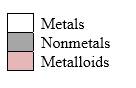
Simplest ion form by each group as follows:
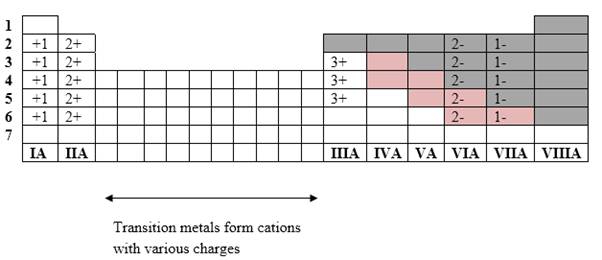
If it’s a compound made by a combination of metal and nonmetal elements, called as ionic compounds.
Ionic chemical compound must have a zero charge. If a compound contains ions;
1. Both positive ions (cations) and negative ions (anions) must be there.
2. No: of cations and anions collectively should produce a zero-net charge.
Answer to Problem 45CR
Explanation of Solution
K and O
(f)
Interpretation:
Predict the simple binary compound that can be formed by the use of electron configuration of the simple ions which are formed by those elements.
Concept Introduction:
Electron configuration describes the positions of electrons of an atom. Depending on that the periodic table is created. Periodic table further defines the simple ion that can be formed by any element. And using those ions, simple binary compounds can be produced.
Order in which orbitals fill to produce the atoms in periodic table as follows:
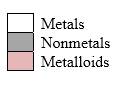
Simplest ion form by each group as follows:
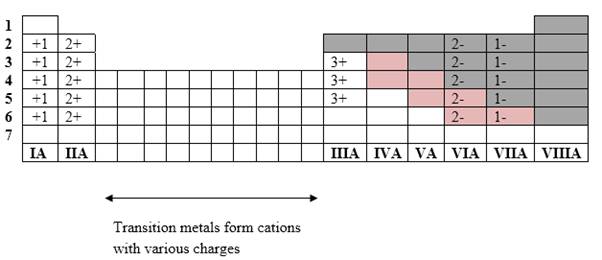
If it’s a compound made by a combination of metal and nonmetal elements, called as ionic compounds.
Ionic chemical compound must have a zero charge. If a compound contains ions;
1. Both positive ions (cations) and negative ions (anions) must be there.
2. No: of cations and anions collectively should produce a zero-net charge.
Answer to Problem 45CR
Explanation of Solution
Sr and Br
( Simple ion =
Want to see more full solutions like this?
Chapter 12 Solutions
Introductory Chemistry: A Foundation - With OwlV2
- Think of forming an ionic compound as three steps (this is a simplification, as with all models): (I) removing an electron from the metal; (2) adding an electron to the nonmetal; and (3) allowing the metal cation and nonmetal anion to come together. a. What is the sign of the energy change for each of these three processes? b. In general, what is the sign of the sum of the first two processes? Use examples to support your answer. c. What must be the sign of the sum of the three process d. Given your answer to part c, why do ionic bonds occur? e. Given your above explanations, why is NaCl stable but not Na2Cl? NaCl2? What about MgO compared to MgO2? Mg2O?arrow_forwardGive the total number of valence electrons in each of the following molecules. msp;a.B2O3c.C2H6Ob.CO2d.NO2arrow_forwardUse data in this chapter (and Chapter 2) to discuss why MgO is an ionic compound but CO is not an ionic compound.arrow_forward
- a. How many sticks did you need to make the skeleton structure?____________ b. How many sticks are left over? ____________ If your model is to obey the octet rule, each ball must have four sticks in it except for hydrogen atom balls, which need and can only have one. Each atom in an octet rule species is surrounded by four pairs of electrons. c. How many holes remain to be filled? ____________ Fill them with the remaining sticks, which represent nonbonding electron pairs. Draw the complete Lewis structure for NH2Cl using lines for bonds and pairs of dots for nonbonding electrons.arrow_forwardhow many shared electrons make up a single covelant bond? what is the maximum number of covalent bonds that can be formed between any two atomsarrow_forwardUse the Lewis model to determine the formula for the compound that forms from each pair of atoms. Ba and Sarrow_forward
- Draw the Lewis symbols for the atoms Al and O. Use the Lewis model to determine the formula for the compound that forms from these two atoms.arrow_forwardA. The Lewis diagram for POCl3 is: The electron-pair geometry around the P atom in POCl3 is ______ There are _____ lone pair(s) around the central atom, so the geometry of POCl3 is ______ B. The Lewis diagram for BH2- is: The electron-pair geometry around the B atom in BH2- is ______ There are _____ lone pair(s) around the central atom, so the geometry of BH2- is _____ (c) The amount of acrylamide found in potato chips is 1.7 mg/kg. If a serving of potato chips is 35 g, how many moles of acrylamide are you consuming? ________molarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole





