
Concept explainers
(a)
Interpretation:
The number of carbon atoms present in the longest chain of following molecule should be determined.
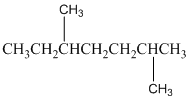
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Saturated hydrocarbon is known as alkane having general molecular formula
According to the rules of naming
(b)
Interpretation:
The number of carbon atoms present in the longest chain of following molecule should be determined.
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Saturated hydrocarbon is known as alkane having general molecular formula
According to the rules of naming alkanes, first rule is to choose the longest continuous chain of carbon atoms known as parent chain and determines the base name of alkane.
(c)
Interpretation:
The number of carbon atoms present in the longest chain of following molecule should be determined.
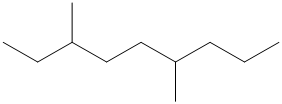
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Saturated hydrocarbon is known as alkane having general molecular formula
According to the rules of naming alkanes, first rule is to choose the longest continuous chain of carbon atoms known as parent chain and determines the base name of alkane.
(d)
Interpretation:
The number of carbon atoms present in the longest chain of following molecule should be determined.
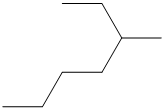
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Saturated hydrocarbon is known as alkane having general molecular formula
According to the rules of naming alkanes, first rule is to choose the longest continuous chain of carbon atoms known as parent chain and determines the base name of alkane.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- What is the meaning of the term tertiary (3) when it is used to classify alcohols? Draw a structural formula for the one tertiary (3) alcohol with the molecular formula C4H10O.arrow_forwardConsider the compound C₂H₃N. Which one of the structures in Figure 4 is the best representation of this compound based on your current knowledge? * A B C D All these structures are good representations of the compound.arrow_forwardIdentify the functional groups in each molecule. . CH₂ CH,CH,CO2 CH;C=H Darvon (analgesic) CH₂N(CH3)2 b. HO CH₂ - တစ CH₂ COOH penicillin G (an antibiotic) ibuprofen (analgesic) .arrow_forward
- 7. What is the molecular formula of griseofulvin? (not condensed structural formula!) OCH 3 O OCH3 H3CO CI H3Carrow_forwardWhich among the following is not an isomer of C5H100? H.arrow_forwardWhat is the commerical compound uses of ethylcyclopentane (C7H14)? Environmental issues? Manufacturing processes?arrow_forward
- 2. Which of the following is not an isomer of the others? a. b. A: A: B: B: C: i D: D: E: O E:arrow_forward1. Name the following molecules H3C H3C H3C a CH₂ ОН d Н b e НО Carrow_forwarddraw the structure and give the systematic name of a compound with molecular formula C5H12 that a. only primary and secondary halogens b. only primary halogens c. one tertiary hydrogensarrow_forward
- F LL 4. Give the indicated name, common or systematic, each of the following compounds. CH₂CH3 common name CH3 OCH 3 common name OH COOH systematic name common name HO3S NH₂ systematic namearrow_forwardHow many isomers can be drawn for C₆H₁₄? A.) 3 B.) 5 C.) 4 D.) 1 E.) 2arrow_forwardDraw the condered structure of the ff. molecular formula a. C6H11Cl b. C4H6 c. C4H8 d. C4H9Farrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





