
Concept explainers
Interpretation:
The relation between ball-and-stick models A and B should be stated along with whether they are constitutional isomers or similar molecules.
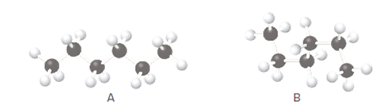
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms.
Saturated hydrocarbon is known as
The compounds having similar chemical formula but different structures are known as isomers.
The isomers which has same molecular formula but arrangement of atoms are different, thus because of different arrangement of atoms,
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- The two molecules drawn below are [Select] options: structural isomers, comforters, or neither conformers nor structural isomers And are [Select] options: the same molecule or different moleculesarrow_forwardConsider 1,2-dimethylcyclohexane.Q: Draw structures for the cis and trans isomers using a hexagon for the sixmembered ring.arrow_forward5) Draw a different constitutional isomer of heptane (C₂H₁6) that contains a quaternary (4°) carbon. Then draw at least 3 other constitutional isomers.arrow_forward
- Question 24 of 28 Fill in the blanks: Write the letter of the pair of compounds that illustrates the given isomerism. H₂C. A m CH3 CH₂ H₂C CH3 Br < Previous H₂N H₂C Positional Isomerism: B TI F B CH₂ CH3 H₂C НО. Br AND G K F CH3 CI CH₂ H₂C. H₂C CH3 D I NH₂ H L OH CH3 CH3arrow_forward12. Identify the set of compounds that have the same functional group. (a) CH, CH,CH, (b) CH, CH,COOH, CH CONH, (e) CH,CH,CHO, H,CO, CH,CH,CH,CH CHO (d) CH,NH, CH,CH,CONH, CH,CH.CONHCH, (e) CHIC(O)CH, CHCOOH, CH,CH CHOarrow_forward7. Functional isomers have unique functional groups but identical reactivity. true or false?arrow_forward
- Draw (zig-zag structure) and name all 75 isomers of decane.arrow_forwardc) The first molecule: trans-2-butene. The second molecule is a mirror image of trans-2-butene. Both molecules are shown on the right. Are the molecules superimposable (identical)? Are the molecules optical isomers (enantiomers)?arrow_forwardDraw the skeletal structures of all isomers of C6H12 that have the (Z)-configuration. Select to Draw an Isomer Select to Draw an Isomer Please select a drawing or reagent from the question areaarrow_forward
- Are these isomers or the same compound?arrow_forward7. Draw structures for compounds having the formula C₂H₂O having the following features (use line-angle structures). a) A compound where ether and alkane are the only functional groups. b) A compound where alcohol and alkane are the only functional groups. c) A compound where there is a carbon-carbon #-bond and an alcohol. d) Two different compounds where there is a carbon-oxygen-bond.arrow_forwardFive structural isomers, or constitutional isomers, have the formula C6H14C6H14. Draw the indicated isomers, grouped by number of carbon atoms in the main chain. a) Draw two isomers with 5 carbon atoms in the main chain. Be sure to include all hydrogen atoms. b) Draw two isomers with 4 carbon atoms in the main chain. Be sure to include all hydrogen atoms.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





