
Concept explainers
(a)
Interpretation:
The resultant product obtained from the metathesis of the alkene
Concept introduction:
Alkene Metathesis Or olefin metathesis
This breaks the double bond of an alkene and then rejoins the fragments. When the fragments are joined, the new double bond is formed between two
Terminal alkene gives the best yields of a single alkene product in metathesis because one of the products is ethane, which is equally removed from the reaction mixture, thus shifting the equilibrium in favor of the other new alkene product.
(b)
Interpretation:
The resultant product obtained from the metathesis of the following alkene should be identified.
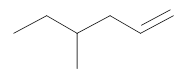
Concept introduction:
Alkene Metathesis Or olefin metathesis
This breaks the double bond of an alkene and then rejoins the fragments. When the fragments are joined, the new double bond is formed between two
Terminal alkene gives the best yields of a single alkene product in metathesis because one of the products is ethane, which is equally removed from the reaction mixture, thus shifting the equilibrium in favor of the other new alkene product.
(c)
Interpretation:
The resultant product obtained from the metathesis of the following alkene should be identified.
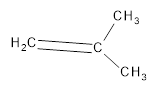
Concept introduction:
Alkene Metathesis Or (olefin metathesis)
This breaks the double bond of an alkene and then rejoins the fragments. When the fragments are joined, the new double bond is formed between two
Terminal alkene gives the best yields of a single alkene product in metathesis because one of the products is ethane, which is equally removed from the reaction mixture, thus shifting the equilibrium in favor of the other new alkene product.
(d)
Interpretation:
The resultant product obtained from the metathesis of the following alkene should be identified.
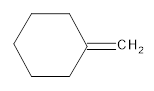
Concept introduction:
Alkene Metathesis Or (olefin metathesis)
This breaks the double bond of an alkene and then rejoins the fragments. When the fragments are joined, the new double bond is formed between two
Terminal alkene gives the best yields of a single alkene product in metathesis because one of the products is ethane, which is equally removed from the reaction mixture, thus shifting the equilibrium in favor of the other new alkene product.
Trending nowThis is a popular solution!

Chapter 12 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- What reagents would you use to prepare each of the following from 3-hexene? a. b.CH3CH2CH2CH2CH2CH3 c. d.arrow_forwardSynthesize the trisubstituted benzene A from benzene.arrow_forward1. Devise a synthesis of the following compounds from benzene. Assume ortho- and para-isomers can be separated. Ноarrow_forward
- .Which one of the following molecules can react as electrophile in reactions with alkenes? CH3OH NaCI HCI KCN NaCI O HCI O KCN O CH3OHarrow_forwardWhat is the major product of electrophilic addition of HBr to the following alkene? Explain your choice.arrow_forwardМСРВА Alkenes are oxidized to give epoxides on treatment with a peroxyacid, RCO3H, such as metachloroperoxybenzoic acid (MCPBA). Peroxyacids transfer an oxygen atom to the alkene with syn stereochemistry, i.e. both C-O bonds form on the same face of the double bond, through a single step mechanism without intermediates. The oxygen atom farthest from the carbonyl group is the one transferred. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions H .CI .CI HOarrow_forward
- In the reaction given below, which of the following is true about the carbocation? CHOICES: It acts as an acid It reacts as an electrophile It undergoes rearrangementarrow_forwardWhich of the following reactions of alkenes takes place with syn stereospecificity? O addition of bromine (treatment with Br2) O oxidation (treatment with OsO4) addition of H2 with Pt O Hydroboration Oxidation O B, C and Darrow_forwardWhat starting alkene reacted with H2O and H2SO4 catalyst is required to produce 2-methylbutan-2-ol?arrow_forward
- Provide synthetic schemes for the followingarrow_forwardWhat alkene with the molecular formula C6H12, when treated with ozone and then dimethyl sulfide, gives the following product(s)?arrow_forwardWhat type of stereochemistry does catalytic hydrogenation afford? Anti-Addition Syn-Addition What is the product of a dissolving metal reduction of an alkyne? An alkyne An alkane OA trans-alkene A cis-alkene Reaction of an alkene with potassium permanganate affords: An anti-diol A syn-diolarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



