
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
7th Edition
ISBN: 9780134240152
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 35P
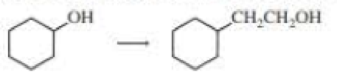
Using the given starting material, any necessary inorganic reagent, and any carbon-containing compounds with no more than two carbons, indicate how each of the following compounds con be prepared:

Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Identify the best reagents to complete the following
reaction.
CI
o
Draw the product(s) formed when A is treated with each reagent.
(a) NaBH4, CH3OH
(b) [1] LiAlH4; [2] H2O
(c) CH3CH2MgBr; [2] H2O
Draw all of the substitution and elimination products formed from the given alkyl halide with each reagent: (a) CH3OH; (b) KOH. Indicate the stereochemistry around the stereogenic centers present in the products, as well as the mechanism by which each product is formed.
Chapter 12 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
Ch. 12.1 - Prob. 1PCh. 12.2 - Which is more reactive an organolithium compound...Ch. 12.2 - Prob. 3PCh. 12.3 - PROBLEM 6♦
Explain why tertiary alkyl halides...Ch. 12.3 - Muscalure is the sex attractant of the common...Ch. 12.3 - Prob. 8PCh. 12.3 - Prob. 9PCh. 12.3 - Prob. 10PCh. 12.4 - Prob. 13PCh. 12.4 - Prob. 14P
Ch. 12.4 - Prob. 15PCh. 12.4 - Show how the Suzuki and/or Heck reactions can be...Ch. 12.4 - Identify two pairs of an alkyl bromide and an...Ch. 12.5 - Prob. 19PCh. 12.5 - Draw the product of ring-closing metathesis for...Ch. 12.5 - Prob. 22PCh. 12 - Prob. 23PCh. 12 - Prob. 24PCh. 12 - Identify A through H.Ch. 12 - 26. Using the given starting material, any...Ch. 12 - Prob. 27PCh. 12 - Prob. 28PCh. 12 - Prob. 29PCh. 12 - Using ethynyleyclohexane as a starting material...Ch. 12 - Prob. 31PCh. 12 - Using the given starting material, any necessary...Ch. 12 - Prob. 33PCh. 12 - A student added an equivalent of...Ch. 12 - Using the given starting material, any necessary...Ch. 12 - Prob. 36PCh. 12 - Prob. 37PCh. 12 - Bombykol is the sex pheromone of the silk moth....Ch. 12 - Prob. 39PCh. 12 - A dibromide loses only one bromine when it reacts...Ch. 12 - What starting material is required in order to...Ch. 12 - Prob. 42PCh. 12 - Prob. 1PCh. 12 - Prob. 2PCh. 12 - Prob. 3PCh. 12 - Prob. 4P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Devise a synthesis for each compound, starting with methylenecyclohexane and any other reagents you need. cyclohexylmethanolarrow_forwardSynthesize the following compound from cyclohexanone and any other organic and inorganic reagents you need.arrow_forwardDetermine the major product from the following reaction: 1) LDA 2) o: 3) Нзоarrow_forward
- Draw the major organic product when this compound is treated with NBS and heat: H3C -CH3arrow_forwardDraw the Substitution and Elimination Reactions:arrow_forwardDraw a stepwise mechanism for the following reaction, one step in the synthesis of the cholesterol-lowering drug pitavastatin, marketed in Japan as a calcium salt under the name Livalo.arrow_forward
- Draw a stepwise mechanism for the following reaction, one step in the synthesis of the cholesterol-lowering drug pitavastatin, marketed in Japan as a calcium salt under the name Livalo.arrow_forward6arrow_forwardWhat product is formed when attached compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4, H2O?arrow_forward
- Which statement is correct? Ethers have a general formula RCOR Ethers are very stable in the basic media O Epoxides are acyclic ethers Ethers may act as acids and bases O Ethers may act only as acidsarrow_forwardWhich of the following sequences of regctions would convert toluene to 2-bromo-4- cydnotoluene? tof Br CN O Nitration, bromination, diazotization, reduction, reaction with cyanide anion OBromination, nitration, reduction, diazotization, reaction with cyanide anion O Bromination, nitration, diazotization, reduction, reaction with cyanide anion O Nitration, bromination, reduction, diazotization, reaction with cyanide anionarrow_forwardAn intoruduction to carbonyl and ketonesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY