
Concept explainers
Core Skill: Connections Look back at Figure 6.3, which describes the hydrolysis of ATP. Why is ATP needed to charge a tRNA?
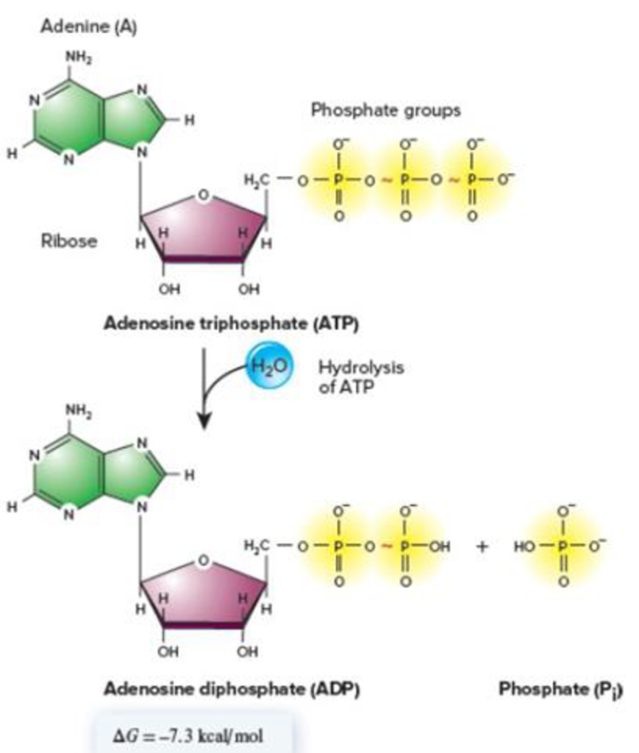
Figure 6.3 The hydrolysis of ATP to ADP and Pi. As shown in this figure, ATP has a net charge of −4, while ADP and Pi are shown with net charges of −2 each. When these compounds are shown in
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Gen Combo Ll Biology; Connect W/learnsmart Labs Access Card
Additional Science Textbook Solutions
Biology: Life on Earth with Physiology (11th Edition)
Campbell Biology in Focus
Microbiology Fundamentals: A Clinical Approach - Standalone book
Principles of Anatomy and Physiology
Microbiology: An Introduction
- VISUALIZE Sketch a pyrimidine nucleotide subunit that would be found only in RNA. Circle and label the three components that make up this type of nucleotide. Explain what changes in the functional groups of this subunit would have to occur for it to be found in a DNA molecule.arrow_forwardneed help to calculate P1 for amino acid Tryptophan. Also please check if I draw the mechanism correctly and completely???arrow_forwardExercise 1 Summarize the data below obtained from the protein experiments for determination of protein concentration in the form of a graph. Plot a graph containing a title and labels for both of the axis. Three readings were taken for each protein concentration. Find the averages and standard deviations for each reading and plot your graph using Microsoft EXCEL, complete with the error bars. This graph represents will be used to measure the protein concentration in an unknown protein solution. calibration curve for a protein assay (next experiment) where this Concentration of protein (Hg/mL) Absorbance, 595 nm 1 2 3 0.04 0.05 0.03 2 0.12 0.11 0.14 5 0.26 0.25 0.25 10 0.49 0.49 0.51 25 0.82 0.85 0.83 50 1.28 1.24 1.25arrow_forward
- Protein purification table: A 50 ml crude skeletal muscle extract contains 32mg of protein per ml. Ten ul of the extract catalyzes a reaction at a rate of 0.14 µmol product per minute. The extract was fractionated by ammonium sulfate precipitation, and the fraction precipitating between 20% and 40% saturation was dissolved in 10ml. The solution contains 50mg/ml protein. Ten ul of this purified fraction catalyzes the reaction at a rate of 0.65 µmol/min (a) What is the degree of purification (fold purification)? (b) What is the percent yield of the enzyme recovered in the purification?arrow_forwardProblem: From the following information determine the amino acid sequence of a peptide. N-terminal Edman gives PTH-Alanine C terminal carboxypeptidase treatment, no observable reaction Trypsin cleavage gives three products Arg Peptide containing Ala, Lys Peptide containing Asp, Met, Phe, Pro Mild Chymotrypsin cleavage gives 2 peptides Peptide containing Asp, Pro Peptide containing Ala, Arg, Lys, Met, Phe CNBr cleavage gives 2 peptides Peptide containing Ala, Arg, Lys and homoserine Peptide containing Asp, Phe, Pro You must supply the answer as the 3-letter amino acid sequence from N-terminus to C-terminus in the form (you must use dashes, not spaces between the amino acids) Met-Thr-Glu-Trparrow_forwardProblem: From the following information determine the amino acid sequence of a peptide. N-terminal Edman gives PTH-Alanine C terminal carboxypeptidase treatment, no observable reaction Trypsin cleavage gives three products Arg Peptide containing Ala, Lys Peptide containing Asp, Met, Phe, Pro Mild Chymotrypsin cleavage gives 2 peptides Peptide containing Asp, Pro Peptide containing Ala, Arg, Lys, Met, Phe CNBr cleavage gives 2 peptide Peptide containing Ala, Arg, Lys and homoserine Peptide containing Asp, Phe, Pro You must supply the answer as the 3-letter amino acid sequence from N-terminus to C-terminus in the form (you must use dashes, not spaces between the amino acids)arrow_forward
- Enzyme Investigation An enzyme was isolated from digestive juices taken from the small intestine. An experiment was set up to test the ability of the enzyme to break down protein. Two test tubes, labeled A and B, were placed in a hot water bath at 37°C, human body temperature. Test tube A contained only protein and test tube B contained protein and the enzyme. The chart below shows the set-up. After two hours, the contents of both test tubes were analyzed. Test tube A showed only the presence of protein. Test tube B showed the presence of the end products of protein digestion, indicating the enzyme had successfully broken down the protein. Identify the end products of protein digestion that made up the contents of test tube B after the two hours.arrow_forwardBased on image of protein (enzyme), need to know: a) Number of amino acids and length (Angstroms) of helix denoted as H2. b) Name the post-translational modification (PTM) the enzyme has underwent. c) Name chemical reagent to undo this PTM and number of equivalents of reagent needed.arrow_forwardcell defense worksheet (1) [Compatibility Mode] Qv Search in Docume Insert Draw Design Layout References Mailings Review View Acrobat A. Century Gothic - 12 A A- U - abe X,2 x² A A Styles Styles Pane Create and Adobe PI Step 13: Take the "Energy and Transport Challenge!" How many ATP did you use? What type(s) of protein(s) were used? Explain when each type was used. Step 14: Take the "Osmosis Challenge!" What is Osmosis? What is the name of the special proieins that let water pass through? Is this passive or active transport? Step 15: From your Scores Sheet record: Lab Score (% correct): Number Correct: Numbar Incen et Page 2 of 3 * English (United States) 505 words E Focus MAR 15 MacBook Air 888 F1 F2 F3 F4 F5 F6 F7 F @ %23 $ & 7 8. Q W T Yarrow_forward
- Bradford technique makes use of the Coomassie blue dye that binds to the protein, with the complex absorbing strongly at 465 nm. O True O Falsearrow_forwardQuestion:- Based on the figure below, predict what peptide bond could be the substrate of each protease(The bond marked in blue is where hydrolysis occurs, choose 2 peptides per protease type) Chymotrypsin:_________ Trypsin:_________ Elastase:_________ 1. SR−SG 2. SF−SG 3. SK−SG 4. SA−SG 5. SV−SG 6. SM−SGarrow_forwardExercise B: Peptide Bond Formation Figure 2 shows two individual amino acids, and then those same two amino acids after they have been linked together by a peptide bond to form a dipeptide. Addition of more amino acids linked by peptide bonds would form a polypeptide, the precursor to a functional protein. нн +| | || H-N-C-Ĉ-0 H. +! | || H-N-C-ċ –0 H. H H H +| | || H-N-C-C-N-C-C- + H CH2 CH2 H CH2 H. CH2 SH SH NH, NH, Figure 2. Formation of a peptide bond between two amino acids. Answer the below questions in your own document. 1. Which two amino acids are shown on the left side of Figure 2? Use the Figure 3.2 from your text to answer this. 2. To which chemical groups do these amino acids belong? 3. Were you able to identify their chemical characteristics based on your rules? If not, you should go back and revise your rules! On the dipeptide shown in Figure 2, label the peptide bond that was formed when the two individual amino acids were joined. Label the free amino and carboxyl…arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
