
ORGANIC CHEMISTRY
6th Edition
ISBN: 9781266633973
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12A, Problem 22P
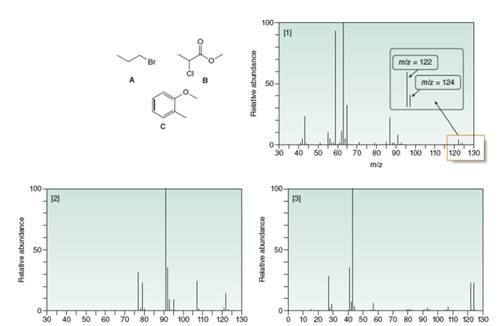
Match each structure to its mass spectrum
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
please help detail the mass spectrum
fill in the table by relating the structure to its mass spec
which compound does this mass spectrum represent?
Please provide only typed answer solution no handwritten solution needed allowed
Chapter 12A Solutions
ORGANIC CHEMISTRY
Ch. 12A.1 - What is the mass of the molecular ion formed from...Ch. 12A.1 - Prob. 3PCh. 12A.2 - Prob. 6PCh. 12A.2 - What molecular ions would you expect for the...Ch. 12A.3 - The mass spectrum of 2,3-dimethylpentane also...Ch. 12A.3 - The base peak in the mass spectrum of 2, 2,...Ch. 12A - Problem-13.24 The mass spectrum of the following...Ch. 12A - Prob. 16PCh. 12A - Which compound gives a molecular ion at m/z= 122,...Ch. 12A - Propose two molecular formulas for each molecular...
Ch. 12A - Propose four possible structures for a hydrocarbon...Ch. 12A - Problem-13.29 What is the molecular formula for...Ch. 12A - Problem-13.30 Propose a molecular formula for rose...Ch. 12A - 13.31 Match each structure to its mass spectrum
Ch. 12A - 13.32 Propose two possible structures for a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
How could you separate a mixture of the following compounds? The reagents available to you are water, either, 1...
Organic Chemistry
During the early part of the 20th century, sulfanilamide (an antibacterial drug) was only administered by injec...
Elementary Principles of Chemical Processes, Binder Ready Version
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Interpret and discuss the mass spectrum given below. Use a table to interpret.arrow_forwardchemistry match the mass spectrum that corresponds to the moleculesarrow_forwardWhat type of compounds/molecules can be detected using LC-MS and what mass range or molecular mass ranges can be detected using LC-MS?arrow_forward
- How can I identify the compound thru this IR an mass spectrum? This is a practice problem and would like to know how to go about this problemarrow_forward5. In a mass spectrum, the peak of greatest abundance is referred to as the_________________________. Relative abundance is the unit along the y-axis ina mass spectrum. What are the units on the x-axis? _____________________.arrow_forwardGg.157.arrow_forward
- The tallest peak in a mass spectrum OA. has the highest charge OB. has the highest isotopic abundance O C. is called the molecular ion O D. is never the molecular ion OE. is called the base peakarrow_forwardPls help ASAP, thank you! "Using the rule of 13, what is a possible formula for M=80 (100%) on a mass spectrum?"arrow_forwardWhat molecular ions will be present in a mass spectrum of 2-chloropropane ? ( HINT: 1. Calculate the molecular weights using each of the common isotopes of Cl. 2. Write down the molecular formula for 2-choloropropane with the isotopes of chlorine and then calculate the mass of molecular ion , m/z).arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY