
(a)
Interpretation:
Three possible structures of given
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of Alkene: An alkene contains
(b)
Interpretation:
Three possible structures of given
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of Alkyne: The general formula of alkyne is
(c)
Interpretation:
Structure of given monosubstituted benzene has to be drawn.
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of monosubstituted benzene: Monosubstituted benzenes are named using ‘-benzene’ as the parent name. The position of one substituent can be located at any one carbon of the benzene ring.
(d)
Interpretation:
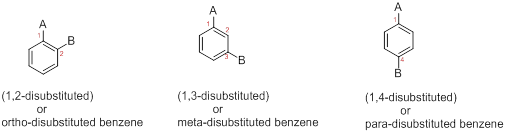
Three possible structures of given disubstituted benzene has to be drawn.
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of Disubstituted benzene: When benzene has more than one substituent, the position of those substituents is indicated by numbers as (1,2), (1,3), and (1,4) or by (1,2-disubstitued) as ortho(o-),(1,3-disubstitued) as meta(m-) or(1,4-disubstitued) as para(p-) in prefix.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Modified Masteringchemistry With Pearson Etext -- Valuepack Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry
- Draw Lewis structures for two compounds of formula C2H7N.arrow_forwardHow many of the carbons in the following structure are stereocenters?arrow_forwardThe explosive trinitrotoluene (TNT) is made by carrying out three successive nitration reactions on toluene. If these nitrations only occur in the ortho and para positions relative to the methyl group, what is the structure of TNT?arrow_forward
- When Alfred Werner was developing the field of coordination chemistry, it was argued by some that the optical activity he observed in the chiral complexes he had prepared was due to the presence of carbon atoms in themolecule. To disprove this argument, Werner synthesized a chiral complex of cobalt that had no carbon atoms in it, and he was able to resolve it into its enantiomers. Design a cobalt(III) complex that would be chiral if it could be synthesized and that contains no carbon atoms.arrow_forwardGive IUPAC names for the five isomers with the formula C6H14.arrow_forwardFor A, B, C, D, E, F, identify the circled functional groups and linkages in the compound in the picture.arrow_forward
- Draw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forwardDraw a Lewis structure for hydroxylamine, NH2OH.arrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. a) d- b) S- c) R- d) I-arrow_forward
- Draw structures corresponding to the following names:(a) 2,4-Dimethylheptan-2-ol(b) 2,2-Diethylcyclohexanol(c) 5-Ethyl-5-methylheptan-1-ol(d) 4-Ethylhexan-2-ol(e) 3-Methoxycyclooctanol(f) 3,3-Dimethylheptane-1,6-diolarrow_forwardDrawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forwardGiven the following choices,a. structure of sterolb. what is the structure of a glycolipid?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





