
Which of the following species will not have permanent dipole moments? (a) Hydrogen cyanide,
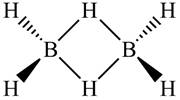
Where the bridging hydrogens are perpendicular to the four terminal hydrogens, which are all coplanar
Trending nowThis is a popular solution!

Chapter 13 Solutions
Physical Chemistry
- What force exists between H2O and CH3CH2OH? Is it Ion-dipole?arrow_forwardIodine and oxygen form a complex series of ions, among them IO4^- and IO5^3-. Draw the Lewis structures for these ions, and specify their electron-pair geometries and the shapes of the ions. What is the hybridization of the I atom in these ions?arrow_forwardDetermine if the following species have permanent dipole moments. a Dichloromethane, CH2Cl2 b Chlorobenzene, C6H5Cl c Ammonia, NH3 d Carbon dioxide, CO2.arrow_forward
- Determine if the following species have permanent dipole moments. a The carbonate ion, CO32 b The phosphate ion, PO43 c Uranium hexafluoride, UF6 d Bromine, Br2.arrow_forwardIdentify These are very weak interactions caused by the momentary changes in electron density in a moleculearrow_forwardBriefly describe how the hydration energy of pentahydrated copper sulphate (CuSO4.5H2O) can be determined. State 2 conditions necessary for the formation of a dative or coordinate bond.arrow_forward
- Which of the following molecules may be polar? (i) pyridine,(ii) nitroethane, (iii) gas-phase BeH2 (linear), (iv) B2H6.arrow_forwardIdentify the bond order and magnetic effect for N2+1.arrow_forwardthe following figure shows the bond distances for the homonuclear diatomics of the 2 nd period p-block elements, as well as those for some of the ions derived from these species. i) Note that N 2 + has a longer bond distance than N 2, but O 2 + has a shorter bond distance than O 2. Explain this difference. ii) Estimate the bond distance of N 2 - and justify your answer.arrow_forward
- Differentiate between Uni-molecular and Bi-molecular?arrow_forwardCalculate the force of attraction between a cation with a valence of +3 and an anion with a valence of -2, the centers of which are separated by a distance of 2.2 nm.arrow_forwardFor the hypothetical cyclic square hydrogen molecular di-cation (H42+), list all electrostatic pairwise interactions including their signs and contributions to stabilising and de-stabilising the molecular structurearrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


