
Concept explainers
a.
Interpretation:
Amine present in given compound has to be classified as primary, secondary or tertiary amine.
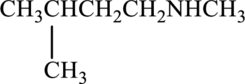
Concept Introduction:
b.
Interpretation:
Amine present in given compound has to be classified as primary, secondary or tertiary amine.
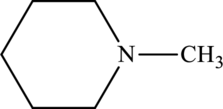
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Classify each of the following as a primary, secondary, or tertiary amine: NH-CH3 a. CH,CH,-N-CH,CH; c. CH3 CH3 CH3 b. CH,-C-NH, ČH3 d.arrow_forward1. Which statement best described the ability of amines to hydrogen bond? A. Primary, secondary and tertiary amines can all hydrogen bond with molecules identical to themselves. B. Primary and secondary amines can hydrogen bond with molecules identical to themselves, but tertiary amines cannot. C. Only primary amines can hydrogen bond with molecules identical to themselves. Secondary and tertiary amines cannot. D. Primary, secondary and tertiary amines cannot hydrogen bond with molecules identical to themselves, but the can hydrogen bond with water.arrow_forwardClassify each amine in the following compounds as 1°, 2°, or 3°. NH2 a. H,N b. CH,CH20. N-CH, spermine (isolated from semen) meperidine (a narcotic) Trade name: Demerolarrow_forward
- The hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forwardWhy are tertiary amines not soluble in water? O a. Primary amine cannot hydrogen bond with water. O b. Amines are non polar compounds. O c. Tertiary amine cannot hydrogen bond with water O d. Secondary amine cannot hydrogen bond with water. Clear my choicearrow_forward19. Which of the following molecules contain an amide? H2N-CHC-OH ČH3 a. c. NH2 NH2 b. H2N H2N-CHC-N-CHC-OH d. CHз CH3arrow_forward
- Match the description to one of the compounds E– H. a. a compound that contains a 1 ° amine and a 1 ° amide b. a compound that contains a 1 ° amine and a 2 ° amide c. a compound that contains a 2 ° amine and a 3 ° amide d. a compound that contains a 3 ° amine and a 3 ° amidearrow_forwardWhich of the following is an amine? Select one: a. HCONH2 b. CH3COCH3 c. CH3F d. CH3NH2arrow_forward11. Classify the following amines as primary (1° ), secondary (2°), or tertiary (3⁰): a. d. NH₂ CH3 | b. CH3-N-CH₂-CH3 CH3-CH₂-N-CH₂-CH2-CH3 | CH3 C. 19 NIHarrow_forward
- Which of the following is NOT a primary amine? Select one: a. N-CH3 b. C. d. NHz 或是 NH₂ ◇ -NHzarrow_forwardDraw a structural formula for each amine and amine derivative. Q.) tert-Butylaminearrow_forwardErythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
