
a.
Interpretation:
Compound that has higher boiling point in the below pair has to be identified.
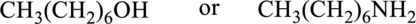
Concept Introduction:
Boiling point of a substance is the temperature at which the liquid phase gets converted into vapor phase. Intermolecular forces plays an important role in the boiling point of a substance. If the intermolecular force is stronger, then the boiling point will be higher. Boiling point of compound that can form hydrogen bonding and polar will be greater than that of the compounds that cannot form hydrogen bonding but can be polar. Boiling point of polar compounds are higher than that of the nonpolar compounds.
b.
Interpretation:
Compound that has higher boiling point in the below pair has to be identified.
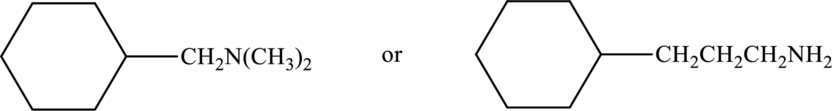
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Give the IUPAC name for each compound. CH3 CH2CH3 Br a. PHCH(CH3)2 b. С. d.arrow_forwardArrange the compounds in order of increasing boiling point. HO. B C D Aarrow_forwardGive the IUPAC name for each compound. OH a. CH3CH(CH₂)4CH3 (select) OH (CH3CH₂)2CHCHCH₂CH3 (select) b. C. d. CH3 OH (select) (select) (select) OH (select) (select) (select)arrow_forward
- THC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body. CH3 OH CH;CH2-OH ethanol CH3 ČH3 (CH),CH3 tetrahydrocannabinol THCarrow_forwardDiethyl ether (CH3CH2OCH2CH3) and butan-1-ol (CH3CH2CH2CH2OH) are constitutional isomers with the same molecular formula, however their boiling points are very different. Which has the higher boiling point? O Diethyl Ether O Butan-1-olarrow_forward1. Simple ketones, like acetone, are often used as industrial solvents for many organically based products such as adhesives and paints. They are considered "universal solvents," because they dissolve so many diverse materials. Explain why these chemicals are good solvents.arrow_forward
- Give the IUPAC name for each structure. Part 1 of 3 CH3(CH2)2CO₂ (CH2)CH3 Part 2 of 3 Part 3 of 3 -CO,—CH,—CH,arrow_forwardName each compound in which the benzene ring is best treated as a substituent. CH3 a. CH3-CH-CH,-CH-CH,-CH–CH,-CH; CH,-CH3 b. CH,-CH-CH=CH-CH,-CH,–CH,-CH, c. CH3-C=C-CH-CH-CH-CH2-CH3 CH3 CH3arrow_forwardClassify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forward
- Acetals are ethers. True or False True Falsearrow_forwardGive the IUPAC name for each structure. Part 1 of 3 CH(CH2),CO,(CH,), CH I Part 2 of 3 Part 3 of 3 CO,—CH, hn Xarrow_forwardWhy is the boiling point of propane-1,3-diol (HOCH2CH2CH2OH) higher than the boiling point of propane-1,2-diol [HOCH2CH(OH)CH3] (215 °C vs. 187 °C)? Why do both diols have a higher boiling point than butan-1-ol (CH3CH2CH2CH2OH, 118 °C)?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





