
Interpretation:
Solid: Solid is a sample of matter that retains its shape and density when not confined. Stronger force of attraction exists between atom/ions/ molecules.
Liquid: A liquid is a sample of matter that conforms to shape of container in which aquires a defined surface in the presence of gravity. In this state weaker force exist between atoms/ ions/ molecules.
Gas: A gas is a sample of matter that conforms to the shape of container in which it is placed and acquires whole volume of container.
Negligible force of attraction exists between atoms/ions/molecules.
Supercritical fluid: Fluid over its critical temperature and pressure exhibiting good solvent power.
Concept used introduction:
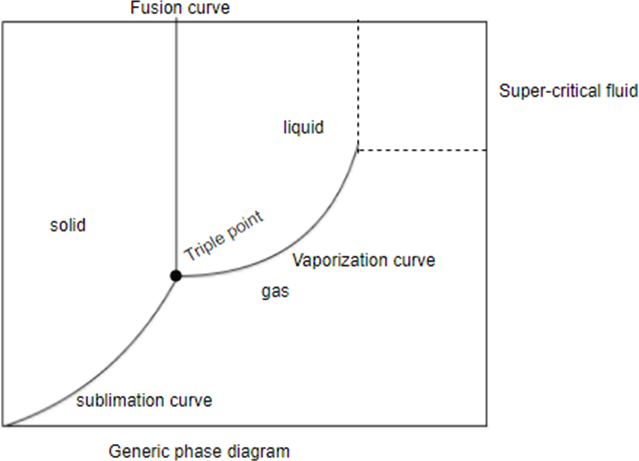
A generic diagram which shows that state of general matter with change in pressure or temperature or both. At a particular temperature or pressure you can find out the state of matter with the help of phase diagram.
To determine:
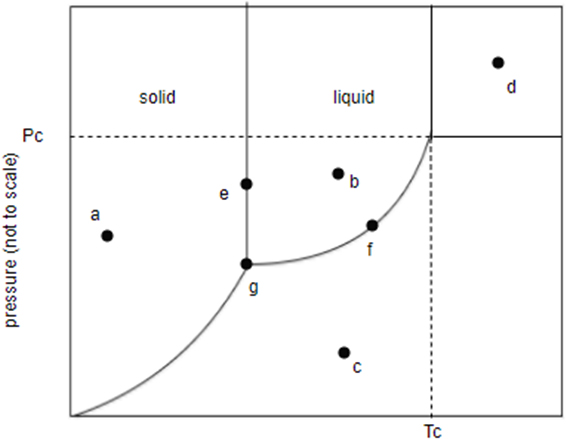
Identify the states present at points through a to g in the phase diagram show here.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for Chemistry: Structure and Properties
- Referring to Figure 9.7, state what phase(s) is/are present at (a) 1 atm, 100C. (b) 0.5 atm, 100C.(c) 0.8 atm. 50C.arrow_forwardReferring to Figure 9.7, state what phase(s) is (are) present at (a) 1 atm, 10C. (b) 3 mm Hg, 20C. (c) 1000 mm Hg, 75C.arrow_forwardDescribe the behavior of a liquid and its vapor in a closed vessel as the temperature increases.arrow_forward
- 8.48 Why must the vapor pressure of a substance be measured only after dynamic equilibrium is established?arrow_forwardWhat feature characterizes the dynamic equilibrium between a liquid and its vapor in a closed container?arrow_forwardA pure substance X has the following properties: Mp=90C, increasing slightly as pressure increases; normal bp=120C; liquid vp=65mm Hg at 100C, 20 mm Hg at the triple point. (a) Draw a phase diagram for X. (b) Label solid, liquid, and vapor regions of the diagram. (c) What changes occur if, at a constant pressure of 100 mm Hg, the temperature is raised from 100C to 150C?arrow_forward
- Use the phase diagram of water in Figure 6.6 and count the total number of phase transitions that are represented.arrow_forwardThe heats of vaporization of liquid O2, liquid Ne, and liquid methanol, CH3OH, are 6.8 kJ/moL 1.8 kJ/mol, and 34.5 kJ/mol, respectively. Are the relative values as you would expect? Explain.arrow_forwardConsider the following data for xenon: Triple point: 121C, 280 torr Normal melting point: 112C Normal boiling point: 107C Which is more dense, Xe(s) or Xe(l)? How do the melting point and boiling point of xenon depend on pressure?arrow_forward
- 8.45 Describe how interactions between molecules affect the vapor pressure of a liquid.arrow_forwardWater at 0C was placed in a dish inside a vessel maintained at low pressure by a vacuum pump. After a quantity of water had evaporated, the remainder froze. If 9.31 g of ice at 0C was obtained, how much liquid water must have evaporated? The heat of fusion of water is 6.01 kJ/mol and its heat of vaporization is 44.9 kJ/mol at 0C.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,  Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





