
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.12E, Problem 13.25P
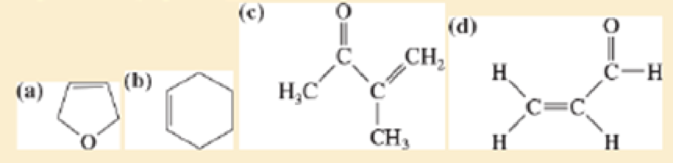
Draw the expected broadband-decoupled 13 C N M R spectra of the following compounds. Use Figure13-41 (page 650) to estimate the chemical shifts.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Spectra data for Unknown Z are listed below. Deduce it's structure. MS: m/z 134 (Molecular Ion) IR: 1720 (strong); 3062 (weak); 2981 (medium); 1605 (medium)
13C NMR and 1H NMR shown on a separate page.
Which of the following will show exactly two peaks in its normal broadband proton decoupled 13C NMR?
Calculate the IHD of C7H6XNO and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.
Chapter 13 Solutions
Organic Chemistry (9th Edition)
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following is the mass spectrum of an unknown compound. The two highest peaks are at m/z 120 and 122. Suggest a structure for this compound. (Data from http://webbook.nist.gov/chemistry/.)arrow_forwardThe electron ionization (EI) mass spectrum of 2-hexanone (C6H12O, molecular weight 100.16) is shown below. Draw the structure responsible for the signal at m/z 58 and name the fragmentation mechanism involved.arrow_forwardCyclohex-2-enone has two protons on its carbon–carbon double bond (labeled Ha and Hb) and two protons on the carbon adjacent to the double bond (labeled Hc). (a) If Jab = 11 Hz and Jbc = 4 Hz, sketch the splitting pattern observed for each proton on the sp2 hybridized carbons. (b) Despite the fact that Ha is located adjacent to an electron-withdrawing C = O, its absorption occurs upfield from the signal due to Hb (6.0 vs. 7.0 ppm). Offer an explanation.arrow_forward
- Please list the relevant 1H-NMR peaks.arrow_forwardCyclohex-2-enone has two protons on its carbon–carbon double bond (labeled Ha and Hb) and two protons on the carbon adjacent to the double bond (labeled Hc). (a) If Jab = 11 Hz and Jbc = 4 Hz, sketch the splitting pattern observed for each proton on the sp2 hybridized carbons. (b) Despite the fact that Ha is located adjacent to an electron-withdrawing C = O, its absorption occurs upeld from the signal due to Hb (6.0 vs. 7.0 ppm). Offer an explanation.arrow_forwardThe number of peaks in the ¹H-NMR spectrum of a com-pound depends on the number of different kinds (environments)of H atoms. How many peaks appear in the spectrum of each iso-mer of C₄H₁₀and of C₅H₁₂arrow_forward
- Why C=O stretching (1680 cm-1) in aromatic ketone is at lower frequency than aliphatic C=O stretch (1715 cm-1)? 1arrow_forwarda) Predict the proton spectrum chemical shift, coupling and integration of the following molecule? vanillin C8H8O3 b) Also predict its cross-peaks in COSY and 1H-13C HETCOR?arrow_forwardWrite notes on the most important absorptions and fragmentations found in the following spectra and use the information to suggest the best possible structure. (Molecular formula: C8H1602, M =144)...arrow_forward
- 6- Below are the figures of two infrared spectra (IR). One corresponds to the spectrum of the cyclohexane compound and the other corresponds to the cyclohexene. Identify each IV with its respective compound and indicate the frequencies and modes of vibration.arrow_forwardDraw the 1H NMR spectrum of the following compounds:-Show all chemical shifts and H assignments.-Show splitting patterns and coupling.-Show integrations.arrow_forwardHow many peaks woudl be observed in the 1H and 13C NMR specutrms of the following compounds?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY