
Organic Chemistry, Books a la Carte Edition (8th Edition)
8th Edition
ISBN: 9780134074580
Author: Bruice, Paula Yurkanis
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.18, Problem 33P
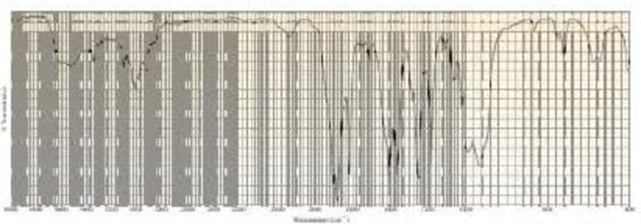
A compound with molecular formula C4H6O gives the infrared spectrum shown here. Identify the compound.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A compound with molecular formula C4H6O gives the infrared spectrum shown here. Identify the compound.
What functional group is evidenced by the IR
spectrum shown?
carboxylic acid
O ketone
O ester
O aldehyde
4. As the energy gap between the HOMO and LUMO orbitals of a molecule decreases, the molecules'
electronic absorption spectrum (UV-vis spectrum) will have a larger Amax (lamba max) value. If
compound X has a Amax = 388 nm, and compound Y has a max = 442 nm, which compound has more
extensive conjugation? Place the name of the compound in the box below.
Chapter 13 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
Ch. 13.1 - Which of the following fragments produced in a...Ch. 13.2 - What distinguishes the mass spectrum of...Ch. 13.2 - What is the most likely m/z value for the base...Ch. 13.3 - Prob. 5PCh. 13.3 - a. Suggest possible molecular formulas for a...Ch. 13.3 - If a compound has a molecular ion with an...Ch. 13.3 - Identify the hydrocarbon that has a molecular ion...Ch. 13.4 - Predict the relative intensities of the molecular...Ch. 13.5 - Which molecular formula has an exact molecular...Ch. 13.5 - Prob. 11P
Ch. 13.6 - Sketch the mass spectrum expected for...Ch. 13.6 - The mass spectra of 1-methoxybutane,...Ch. 13.6 - Primary alcohols have a strong peak at m/z = 31....Ch. 13.6 - Identify the ketones responsible for the mass...Ch. 13.6 - Prob. 16PCh. 13.6 - Using curved arrows, show the principal fragments...Ch. 13.6 - The reaction of (Z)-2-pentene with water and a...Ch. 13.9 - a. Which is higher in energy: electromagnetic...Ch. 13.9 - Prob. 20PCh. 13.13 - Prob. 21PCh. 13.14 - Which occur at a larger wavenumber: a. the C O...Ch. 13.14 - Prob. 23PCh. 13.14 - Prob. 24PCh. 13.14 - Rank the following compounds from highest...Ch. 13.14 - Which shows an O H stretch at a larger...Ch. 13.16 - Prob. 27PCh. 13.16 - a. An oxygen-containing compound shows an...Ch. 13.16 - Prob. 29PCh. 13.16 - For each of the following pair of compounds, name...Ch. 13.17 - Which of the following compounds has a vibration...Ch. 13.17 - Prob. 32PCh. 13.18 - A compound with molecular formula C4H6O gives the...Ch. 13.20 - Prob. 34PCh. 13.20 - Prob. 35PCh. 13.21 - Predict the max of the following compound:Ch. 13.21 - Prob. 37PCh. 13.23 - a. At pH = 7 one of the ions shown here is purple...Ch. 13.23 - Prob. 39PCh. 13.23 - Prob. 40PCh. 13 - In the mass spectrum of the following compounds,...Ch. 13 - Prob. 42PCh. 13 - Draw structures for a saturated hydrocarbon that...Ch. 13 - Rank the following compounds in order of...Ch. 13 - For each of the following pairs of compounds,...Ch. 13 - a. How could you use IR spectroscopy to determine...Ch. 13 - Assuming that the force constant is approximately...Ch. 13 - Norlutin and Enovid are ketones that suppress so...Ch. 13 - In the following boxes, list the types of bonds...Ch. 13 - A mass spectrum shows significant peaks at m/z. =...Ch. 13 - Prob. 51PCh. 13 - Prob. 52PCh. 13 - Prob. 53PCh. 13 - The IR spectrum of a compound with molecular...Ch. 13 - Rank the following compounds from highest...Ch. 13 - Rank the following compounds from highest...Ch. 13 - What peaks in their mass spectra can be used to...Ch. 13 - Prob. 58PCh. 13 - Which one of the following five compounds produced...Ch. 13 - Prob. 60PCh. 13 - Each of the IR spectra shown below is accompanied...Ch. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - How can IR spectroscopy distinguish between...Ch. 13 - Prob. 65PCh. 13 - Prob. 66PCh. 13 - Give approximate wavenumbers for the major...Ch. 13 - Prob. 68PCh. 13 - Which one of the following live compounds produced...Ch. 13 - Phenolphthalein is an acid-base indicator. In...Ch. 13 - Prob. 71PCh. 13 - How can you use UV spectroscopy to distinguish...Ch. 13 - Prob. 73PCh. 13 - The IR and mass spectra for three different...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A ¹H NMR spectrum is shown for a molecule with the molecular formula of C9H10O2. Draw the structure that best fits this data. 1H 11 10 9 8 SH 7 4 214 21 1 pom Qarrow_forward2. The full structural formulae of three organic compounds, P, Q and R, are shown below. H H H H HH HHHH Н-С -с- Н H-C- C - C = C – H H - C - C = C – C – H H H H H Q P (a) State one similarity between P, Q and R in terms of their molecular formulae. (b) Name the homologous series that compounds P, Q and R belong to. (c) State one similarity between Q and R in terms of chemical bonding id) Which of these compounds are isomers? Explain your answer.arrow_forwardwhich of the following compounds absorb the radiation at longer wave length, Why ? 1- H;C-CH CH-CH=CH-CH3 2- H,C CH CH-CH=CH-C-CH3arrow_forward
- Compound 1 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Compound 2 has molecular formula C7H15Br. It shows two signals in the 1H-NMR spectrum, one at 1.08 ppm and one at 1.59 ppm. The relative integrals of these two signals are 3 and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardGive the relationship between pairs A and C below. The possible relationship are: "same compound", "constitutional (structural) isomers", "cis-trans isomers", and "not isomers". A and Br Br Br В and H Br C andarrow_forwardYou have the following solutions, A-E. Which solution has the highest absorbance value at 280 nm? Solution A: 1.0 mM Gly-Ser Solution B: 10 mM Gly-Ser Solution C: 1.0 mM Phe-Ser Solution D: 1.0 mM Tyr-Ser Solution E: 1.0 mM Trp-Ser Solution C Solution E Solution B Solution D Solution Aarrow_forward
- Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardThe following NMR and IR spectra were obtained for a molecule with molecular formula C2H4O2. Draw the structure of the molecule that is consistent with the two spectra. Note. In the NMR spectrum, s stands for singlet. LOD C2H,O2 3H, s 3000 cm-1 1H, s 1720 cm-1 D 4000 3000 2000 1500 1000 HAVENUMBERI l 12 10 8 4 -2arrow_forwardHow do the three isomers of molecular formula C3H6O (A, B, and C) differ in their IR spectra?arrow_forward
- helparrow_forwardSee attached picture Question.) Below are the NMR spectra and molecular formulas of three molecules. Draw the complete structure in the boxes below. All the atoms in each molecule are closed shell (octets) and uncharged. Lack of IR data does not mean that a specific functional group is not present in the molecule.arrow_forward1) Consider the following 5 compounds: NH2 H3C- NHh = white Cos H=red NH2 NH2 NH2 COOH H- -COOH COOH H3C čOOH NH2 čOOH H3C H- -NH2 ČOOH II II H3C H NH2 COOH H, COOH H3C. COOH NH2 NH2 COOH NH2 IV Indicate which of the following TWO structures are described by the following (use all ten a. possible combinations, +&4, I & II, &-V, 1& V, I & I,tt&V, H&V, II & IV, II & V, V-& Identical:+V, Enantiomers: | V,1-11 Diastereomers: lIV IV, N V b. Indicate which of the above compounds WILL rotate plane polarized light when dissolved in solution by writing their Roman numeral(s) here.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY