
Concept explainers
(a)
Interpretation:
The major product of the reaction of 2-methyl-2-butene with
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bond strength is depends on the formation of the radical, if the radical is involving in resonance which is weakest bond strength.
Peroxide is a radical initiator, it has O-O bond in the molecule.
It is used in the radical reaction for the radical initiator.
Markovnikov’s rule:
(b)
Interpretation:
The major product of the reaction of 2-methyl-2-butene with
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bond strength is depends on the formation of the radical, if the radical is involving in resonance which is weakest bond strength.
Peroxide is a radical initiator, it has O-O bond in the molecule.
It is used in the radical reaction for the radical initiator.
Markovnikov’s rule:
Alkene undergoes hydrohalogenation using hydrogen halide through breaking of carbon to carbon double bond, followed by the electrophilic addition of a hydrogen atom and halogen. The halide ion will add to the more substituted carbon in the alkene.
(c)
Interpretation:
The major product of the reaction of 2-methyl-2-butene with
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bond strength is depends on the formation of the radical, if the radical is involving in resonance which is weakest bond strength.
Peroxide is a radical initiator, it has O-O bond in the molecule.
It is used in the radical reaction for the radical initiator.
Bromination of alkene in presence of peroxide:
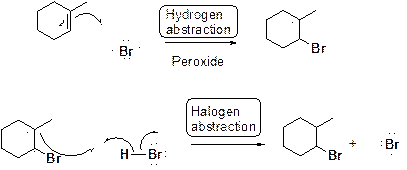
(d)
Interpretation:
The major product of the reaction of 2-methyl-2-butene with
Concept introduction:
Radical or free radical: unpaired valence electron of an atom, molecule, or ion is called as radical.
Bond strength is depends on the formation of the radical, if the radical is involving in resonance which is weakest bond strength.
Peroxide is a radical initiator, it has O-O bond in the molecule.
It is used in the radical reaction for the radical initiator.
Chlorination or iodination of alkene in presence of peroxide:
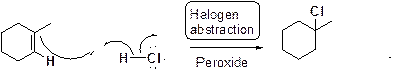
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Draw structural formulas for the alkene that gives each alcohol upon hydroborationoxidation. OH HO, b. a.arrow_forwardDraw the major products obtained from the reaction of one equivalent of HCl with the following compounds. For each reaction, indicate the kinetic andthermodynamic products. a. 2,3-dimethyl-1,3-pentadiene b. 2,4-dimethyl-1,3-pentadienearrow_forwardWhat is the major organic product obtained from the following reaction? A. aldehyde B. alcohol C. anhydride D. carboxylic acidarrow_forward
- 4. What is the major organic product obtained from the following reaction? 1. NaNH, 2. CH₂CH₂Br + 0 5. What is the major organic product obtained from the following reaction? 1. BH3 2. H₂O₂, NaOHarrow_forwardProvide the reagents necessary to carry out the following transformations. Some of these may be multiple steps syntheses and may involve the use of other organic molecules. a. b. CH₂ OH Ph OHarrow_forward11. What is the major organic product obtained from the following reaction? CH₂CH₂C=CH A. 1 B. 2 C. 3 D. 4 H₂O HgSO4 H₂SO4 1 OH 3 OH OH 2 OH 4arrow_forward
- What is the major organic product obtained from the following reaction? A. B. OH COOH CI 1. Mg 2. CO2 3. H3O+ C. D. CH₂OH CHOarrow_forwardAlkyl halide can be prepared from the addition of H-X to alkenes. Identify the product in the reaction. H-Br Select one: a. 1,2-dibromo-2-methylpropane b. 1-bromo-2-methylpropane c. t-butyl bromide (or 2-bromo-2-methylpropane) d. 2-methylpropanearrow_forwardWhat is the IUPAC name for the following substituted benzene? NO2 Br NO2 Select one: O a. 1-bromo-2,5-dinitrobenzene O b. 3-bromo-1,4-dinitrobenzene O c. p-dinitro-2-bromo-benzene O d. 2-bromo-1,4-dinitrobenzene O e. 1-bromo-3,6-dinitrobenzenearrow_forward
- 9. What is the major organic product obtained from the following reaction? 1. BH3 2. H₂O₂, NaOHarrow_forwardWhat is the major organic product obtained from the following reaction? A. B. OH OH 1.2 moles 2. H3O* C. MgBr هذه معه OHarrow_forward40. Deuterium oxide, D20, is water that contains the stable hydrogen isotope 2H, usually shown as D. When dissolved in deuterium oxide, ethanol undergoes the following reaction: * CH3CH2OH + D½0 → CH;CH2OD + HOD What is the best way of showing that this reaction has occurred? A. By testing the product with phosphorus pentachloride and not getting steamy fumes. B. By determining the mass spectrum of the mixture and showing peaks at m/z values of 20 and 46. C. By fractionally distilling the mixture and determining the amount of CH3CH2OD using a Geiger counter. D. By determining the mass spectrum of the mixture and showing peaks at m/z values by 19 and 47. A В O Darrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





