
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781337598255
Author: Spencer L. Seager
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 14.39E
Fructose, present with glucose in honey, reacts with Benedict’s reagent. Circle the structural features that enable fructose to react.
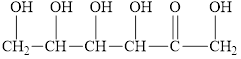
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Identify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. ketohexoseb. alcohol hexosec. aldohexosed. carboxylic acid pentosee. deoxyhexoseThe product is a(n)a. alcohol pyranosesb. etherc. ester d. hemiacetals (anomers)e. acetals (disaccharide)The reaction type is:a. hemiacetal formationb. mutarotationc. hydrolysisd. oxidation (benedict's)e. hemiacetal formationf. reduction (hydrogenation)
Classify the following disaccharide.
a. beta 1-2 disaccharide
b. alpha 1-4 disaccharide
c. alpha 1-6 disaccharide
d. alpha 1-2 disaccharide
e. beta 1-4 disaccharide
f. beta 1-6 disaccharide
What functional groups are present in a carbohydrate molecule?
a. Carboxyl and carbonyl groups
b. Alcohol and carboxyl groups
c. Hydroxyl and carbonyl groups
d. Hydroxyl and hydrogen groups
Chapter 14 Solutions
Bundle: Chemistry for Today: General, Organic, and Biochemistry, Loose-Leaf Version, 9th + LMS Integrated OWLv2, 4 terms (24 months) Printed Access Card
Ch. 14 - Prob. 14.1ECh. 14 - Prob. 14.2ECh. 14 - Identify each of the following compounds as an...Ch. 14 - Identify each of the following compounds as an...Ch. 14 - Prob. 14.5ECh. 14 - Prob. 14.6ECh. 14 - Prob. 14.7ECh. 14 - Prob. 14.8ECh. 14 - Draw structural formulas and give IUPAC names for...Ch. 14 - Draw structural formulas and give IUPAC names for...
Ch. 14 - Each of the following names is wrong. Give the...Ch. 14 - Each of the following names is wrong. Give the...Ch. 14 - Prob. 14.13ECh. 14 - Prob. 14.14ECh. 14 - Prob. 14.15ECh. 14 - Explain why propane boils at 42C, whereas ethanal,...Ch. 14 - Use a dotted line to show hydrogen bonding between...Ch. 14 - Use a dotted line to show hydrogen bonding between...Ch. 14 - Prob. 14.19ECh. 14 - Prob. 14.20ECh. 14 - Prob. 14.21ECh. 14 - Prob. 14.22ECh. 14 - Prob. 14.23ECh. 14 - Prob. 14.24ECh. 14 - Label each of the following as acetals, ketals, or...Ch. 14 - Label each of the following as acetals, ketals, or...Ch. 14 - Label each of the following structures as a cyclic...Ch. 14 - Label each of the following structures as a...Ch. 14 - What two functional groups react to form the...Ch. 14 - Hemiacetals are sometimes referred to as potential...Ch. 14 - Complete the following statements: a. Oxidation of...Ch. 14 - Prob. 14.32ECh. 14 - Prob. 14.33ECh. 14 - Prob. 14.34ECh. 14 - Prob. 14.35ECh. 14 - Not all aldehyde give a positve Bendicts test....Ch. 14 - A stockroom assistant prepares three bottles, each...Ch. 14 - Glucose, the sugar present within the blood, gives...Ch. 14 - Fructose, present with glucose in honey, reacts...Ch. 14 - Prob. 14.40ECh. 14 - Prob. 14.41ECh. 14 - Complete the following equations. If no reaction...Ch. 14 - Complete the following equations. If no reaction...Ch. 14 - Describe the products that result when hydrogen...Ch. 14 - Prob. 14.45ECh. 14 - Draw structural formulas for the products of the...Ch. 14 - The following compounds are cyclic acetals or...Ch. 14 - The following compounds are cyclic acetals or...Ch. 14 - Write equations to show how the following...Ch. 14 - Prob. 14.50ECh. 14 - Identify the most important aldehyde and ketone...Ch. 14 - Using Table 14.3, name an aldehyde or ketone used...Ch. 14 - Prob. 14.53ECh. 14 - CH3COH(O)CH3COOHacetaldehydeaceticacid You need to...Ch. 14 - The addition of water to aldehydes and ketones...Ch. 14 - Prob. 14.56ECh. 14 - Formaldehyde levels above 0.10mg/1000L of ambient...Ch. 14 - In the IUPAC name for the following ketone, it is...Ch. 14 - Why can formaldehyde (CH2O) be prepared in the...Ch. 14 - Other addition reactions of aldehydes occur....Ch. 14 - Prob. 14.61ECh. 14 - Prob. 14.62ECh. 14 - Vanilla flavoring is either extracted from a...Ch. 14 - Prob. 14.64ECh. 14 - The use of acetone in laboratory experiments must...Ch. 14 - Prob. 14.66ECh. 14 - Prob. 14.67ECh. 14 - Which of the following would be classified as a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Glucose, the sugar present within the blood, gives a positive Benedicts test. Circle the structural features that enable glucose to react.arrow_forwardFats belong to the class of organic compounds represented by the general formula, RCOOR', where R and R' represent hydrocarbon groups; therefore, fats are: a. ethers. b. soaps. c. esters. d. lipases.arrow_forwardPlease answer all subparts pertaining to question 5arrow_forward
- What is the name of the alcohol produced by the fermentation of sugars and starches and container in alcoholic beverages A.Methyl alcohol B.Ethanol C. Isopropyl alcohol D. Methanol E. Propanolarrow_forwarda) Aldehydes are easily converted (oxidised) to form carboxylic acids.Ketones, on the other hand, do not oxidise to form carboxylic acids.Suggest a reason why this is so.arrow_forwardTollen’s Test: (Silver Mirror Test) For Reducing SugarsSummarize the results of the Tollen's test withA. GlucoseB. LactoseC. SucroseD. Starcharrow_forward
- Identify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. two hemiacetalsb. disaccharidec. esterd. ethere. alcohol pyranosesThe product is a(n)a. beta 1-6 disaccharideb. beta 1-4 disaccharidec. alpha 1-6 disaccharided. beta 1-2 disaccharidee. alpha 1-2 disaccharidef. alcohol pyranosesg. alpha 1-4 disaccharideh. two hemiacetalsThe reaction type is:a. hydrolysisb. mutarotationc. reduction (hydrogenation)d. hemiacetal formatione. acetal formationf. oxidation (benedict's)arrow_forwardDISACCHARIDE The condensation of two monosaccharide forms a disaccharide or polysaccharide if several monosaccharides are involved. When monosaccharides join together one water molecule is produced. Usually the C-1 OH and C-4 OH or C-1 OH and C-6 OH are involved in the formation of new bond called the glycosidic bond (an ether linkage). Two possible glycosidic bonds are the 1,4’-alpha glycosidic bond and 1,4’-beta glycosidic bond. Did you notice the difference between the two glycosidic bonds? 1,4'-alpha glycosidic bond ÇH2OH ÇH2OH Он H. H OH is lost -H2O H H он CH2OH Он Но OH Но + ÇH2OH H OH ОН alpha anomer он H H OH Он H OH H is lost>HỌ OH 1,4'-alpha glycosidic bond OH alpha anomer 1,4'-beta glycosidic bond beta anomer CH2OH OH ÇH2OH H is removed H Он О он CH2OH НО CH2OH OH H -O OH H OH H H H2O H OH OH OH но H OH is removed Но H OH OH 1,4'-beta glycosidic bond beta anomer -오arrow_forwardIdentify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. ketopentoseb. aldopentosec. ketotriosed. alcohol pentosee. carboxylic acid tetroseThe product is a(n)a. aldohexosesb. alcohol pentosesc. carboxylic acid pentosesd. deoxypentosee. ketopentosesThe reaction type is:a. hemiacetal formationb. reduction (hydrogenation)c. hydrolysisd. acetal formatione. mutarotationf. oxidation (benedict's)arrow_forward
- Directions: Answer the following questions briefly. Write your answer on the space provided after the question. Sara is studying nutrition and as an assignment she has been asked to find examples of each type of carbohydrate in the food that she consumes over the course of a week. Sarah is eating pasta today and knows that it contains starch made of a long chain of many sugars. What type of carbohydrate would you classify starch as? Support your answer. Malik has a huge track meet tomorrow. Malik should focus on eating foods rich in what type of macromolecule? Why?arrow_forwardMultiple Choice: Select the correct answer from the given choices. Write your answer on a separate sheet of paper. 1. Carbohydrates are also known as A. Carbonates B. Glycolipids C.Hydrates of carbon 2. Which of the following class of carbohydrate, which cannot be hydrolysed? D. Polysaccharides A. Disaccharides B. Monosaccharide C. Polysaccharides D. Proteoglycan 3. Chitin consists of A. D-glucose units B. N-acetyl glucosamine C. N-acetyl muramic acid D. N-acetyl muramic acid and N-acetyl glucosamine 4. Which of the following is the component of starch ? A. branched amylose and branched amylopectin B. branched amylose and unbranched amylopectin C. unbranched amylose and branched amylopectin D. unbranched amylose and unbranched amylopectin 5. Lactose is a disaccharide consists of A. glucose and fructose C. glucose and ribose B. glucose and galactose D. glucose and sucrose 6. Sucrose is a A. disaccharide B. monosaccharide C. polysaccharide D. triose 7. In polysaccharides,…arrow_forwardDraw the structure of the product that forms when the carbonyl compound shown is treated with K,Cr₂O, If no reaction occurs, draw the structure of the organic starting material (reactant). CH,(CH,), (CO)CH(CH,), Click and drag to start drawing a structure. 1 :0 Statover G 0arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY