
Concept explainers
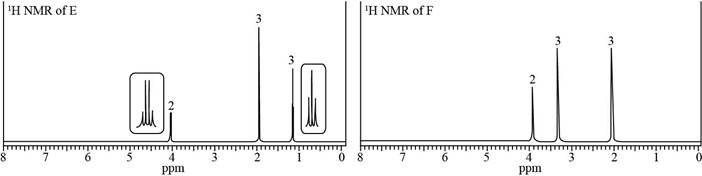
Identify the structures of isomers E and F (molecular formula
each signal.
a. Compound E:IR absorption at
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
ORG CHEM LL W/ LL SG&CONPLUS PKG>IC<
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Inorganic Chemistry
Chemistry & Chemical Reactivity
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Chemistry by OpenStax (2015-05-04)
Elementary Principles of Chemical Processes, Binder Ready Version
- When measured on a spectrometer operating at 200 MHz, chloroform (CHCl3) shows a single sharp absorption at 7.3 δ. (a) How many parts per million downfield from TMS does chloroform absorb? (b) How many hertz downfield from TMS would chloroform absorb if the measurement were carried out on a spectrometer operating at 360 MHz? (c) What would be the position of the chloroform absorption in δ units when measured on a 360 MHz spectrometer?arrow_forward3-Bromo-1-phenyl-1-propene shows a complex NMR spectrum in which the vinylic proton at C2 is coupled with both the C1 vinylic proton (J = 16 Hz) and the C3 methylene protons (J = 8 Hz). Draw a tree diagram for the C2 proton signal, and account for the fact that a five-line multiplet is observed.arrow_forwardHow many signals are greater than 100 ppm in the 13C NMR? How many signals are less than 100 ppm in the 13C NMR? How many signals total in the 13C NMR?arrow_forward
- 1. 1H-NMR spectrum of 1,3-propanediol (HO-CH2-CH2-CH2-OH) shows a quintet at 1.81 ppm, a singlet at 2.75 pm, and a triplet at 3.83 ppm. Assign each signal to the protons it corresponds to in the molecule. Explain the splitting pattern observed for each signal.arrow_forwardWhich compounds (A-F) match with the corresponding IR spectrums (1-2)?arrow_forwardHow many signals are greater than 100 ppm in the 13C NMR? How many signals are less than 100 ppm in the 13C NMR? How many total signals in the 13C NMR?arrow_forward
- An unknown compound (x) contains only carbon and hydrogen, has MW=112 and exhibits the spectral data below. In addition to the IR signal listed below, there are only peaks corresponding to C-H stretches (between 3300 and 2900) and several in the "fingerprint region". IR (cm-1) 2145 CNMR (ppm) 77.8, 70.1, 30.2 H NMR (ppm) 2.45 (singlet)arrow_forwardThe mass spectrum of Compound H, with a molecular formula C8H9NO is shown below. 1.Calculate the degree of unsaturation of Compound H. What information can be obtained from this calculated value? 2.Draw the most likely species responsible for the signals at the base peak. 3.Identify and draw the structure of Compound H.arrow_forwardBelow are three MS spectra, Spectra 1, 2, and 3. Each of this mass spectrum corresponds to either Compound A (contains one Cl in the molecular formula), Compound B (one I in the molecular formula), and Compound C (one Br in the molecular formula). Identify which spectra corresponds to which compound. Provide explanations for your choices.arrow_forward
- The mass spectrum of a ketone (C7H14O) exhibits a base peak at m/z = 57. Is the ketone most likely heptan-2-one, heptan-3-one, or heptan-4-one? Explainarrow_forwardProvide structures that are consistent with the attached DEPT spectra (A-C). Each set of spectra includes a DEPT-90 (top), DEPT-135 (middle) and standard 13C decoupled spectrum (bottom). a) C5H11Brarrow_forwardLook at the structure of ethyl acetate in your notebook. In which region of the 13C spectrum of ethyl acetate would you NOT expect any peaks? a.0 - 50 ppm b.50 - 100 ppm c.100 - 160 ppm d.160 - 220 ppmarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

