
Concept explainers
Identify the structures of isomers H and I (molecular formula
a. Compound H: IR absorptions at
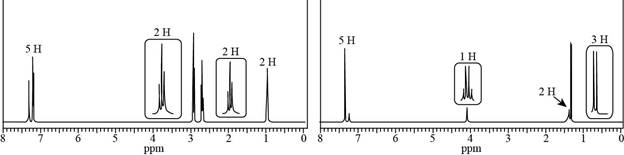
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
ORG CHEM LL W/ LL SG&CONPLUS PKG>IC<
Additional Science Textbook Solutions
Basic Chemistry
Chemistry For Changing Times (14th Edition)
Chemistry: The Central Science (14th Edition)
Organic Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- Treatment of compound E (molecular formula C4H8O2) with excessCH3CH2MgBr yields compound F (molecular formula C6H14O) afterprotonation with H2O. E shows a strong absorption in its IR spectrum at1743 cm−1. F shows a strong IR absorption at 3600−3200 cm−1. The 1HNMR spectral data of E and F are given. What are the structures of E andF?Compound E signals at 1.2 (triplet, 3 H), 2.0 (singlet, 3 H), and 4.1 (quartet, 2 H) ppm Compound F signals at 0.9 (triplet, 6 H), 1.1 (singlet, 3 H), 1.5 (quartet, 4H), and 1.55 (singlet, 1 H) ppmarrow_forwardIdentify the structures of isomers H and I (molecular formula C8H11N).a.Compound H: IR absorptions at 3365, 3284, 3026, 2932, 1603, and 1497 cm−1b.Compound I: IR absorptions at 3367, 3286, 3027, 2962, 1604, and 1492 cm−1arrow_forwardCompounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forward
- Propose a structure consistent with each set of data. C10H14: IR absorptions at 3150–2850, 1600, and 1500 cm−1arrow_forwardPart 3A Set 1. Can IR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. What significant absorptions would be observed in the IR spectrum?arrow_forward1. Deduce a possible structure for the compound with the IR absorptions below. (a) C5H8O: 2950, 1750 cm-1 (b) C4H8O: 2950, 2820, 2715, 1715 cm-1 2. How could IR spectroscopy be used to distinguish between the following pair of compounds? (a) CH2=CHCH2CH(CH3)2 and CH3CH2CH2CH(CH3)2 (b) CH3OCH2CH3 and CH3CH2CH2OH 4. How does the O-H stretch in the IR spectrum of a carboxylic acid differ from the O-H stretch of an alcohol?arrow_forward
- A compound shows strong, very broad IR absorption in the region 3300–3600 cm-1 and strong, sharp absorption at 1715 cm-1. What functional group accounts for both of these absorptions?arrow_forwardIdentify the structures of D and E, isomers of molecular formula C6H12O2, from their IR and 1H NMR data. Signals at 1.35 and 1.60 ppm in the 1H NMR spectrum of D and 1.90 ppm in the 1H NMR spectrum of Eare multiplets.a. IR absorption for D at 1743 cm−1b. IR absorption for E at 1746 cm−1arrow_forwardAssume that you have a compound with the formula C4H8O. a) How many double bonds and/or rings does your compound contain? b) If your compound shows an infrared absorption peak at 1715 cm-1, what functional group does it have? c) If your compound shows a single 1H NMR absorption peak at 2.1 δ, what is its structure?arrow_forward
- Draw a structure consistent with the following set of data: a hydrocarbon with a molecular ion at m/z = 68 and IR absorptions at 3310, 3000−2850, and 2120cm−1.arrow_forwardcompound with the molecular formula C7H9N exhibits IR bands at 3450 cm-1 (medium, doublet), and 855 cm-1 (strong) and shows the following major mass spectral signals (m/z): 106 (base); 107 (M+, about 70% of base), 91 (40% of base), and 77 (about 20% of base). Deduce a reasonable structure from this data.arrow_forward1.2 A compound with molecular formula C3H6O consists of major peaks at 2950, 1720,and 1400 cm-1 of an IR spectrum. Assign the peaks and draw a structure for themolecule that might be the compound.(5)1.3 Show how you would use infrared spectroscopy to distinguish between cyclohexane,cyclohexanol and cyclohexanone.(6)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
