
(a)
Interpretation:
The given compound has to be classified as an ether, an alcohol, or neither an ether or alcohol.
Concept Introduction:
Ether is an organic compound. In ether, an oxygen atom is bonded to two carbon atoms by a single bond. The groups that are attached to the carbon atom by a single bond may be alkyl, aryl, or cycloalkyl groups. The functional group of ether is
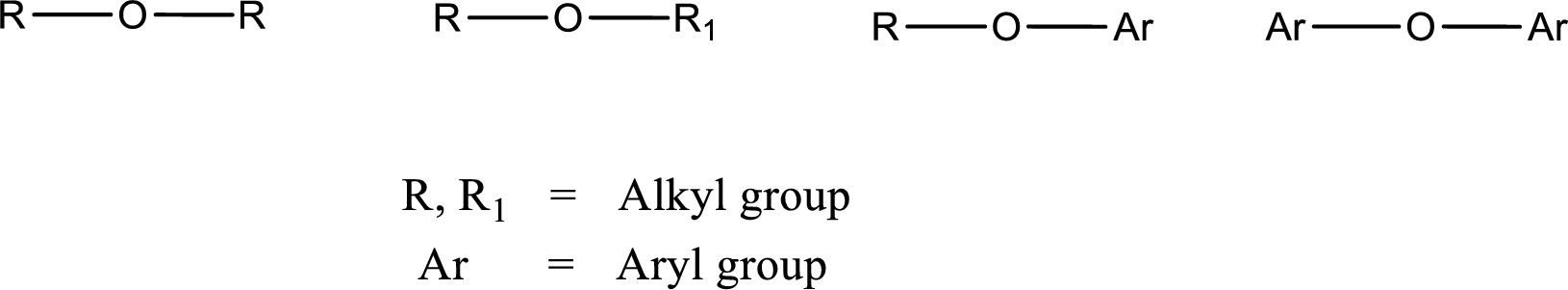
Alcohol is a compound in which a hydrocarbon group is bonded to a hydroxyl group. The generalized formula for alcohol can be given as,
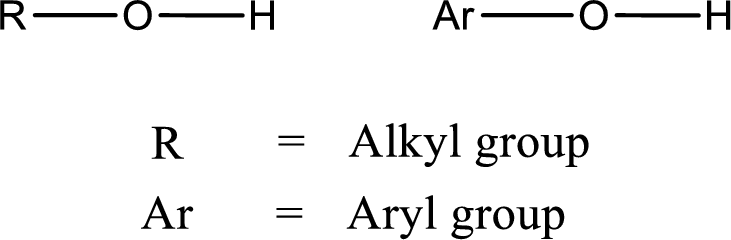
(b)
Interpretation:
The given compound has to be classified as an ether, an alcohol, or neither an ether or alcohol.
Concept Introduction:
Ether is an organic compound. In ether, an oxygen atom is bonded to two carbon atoms by a single bond. The groups that are attached to the carbon atom by a single bond may be alkyl, aryl, or cycloalkyl groups. The functional group of ether is
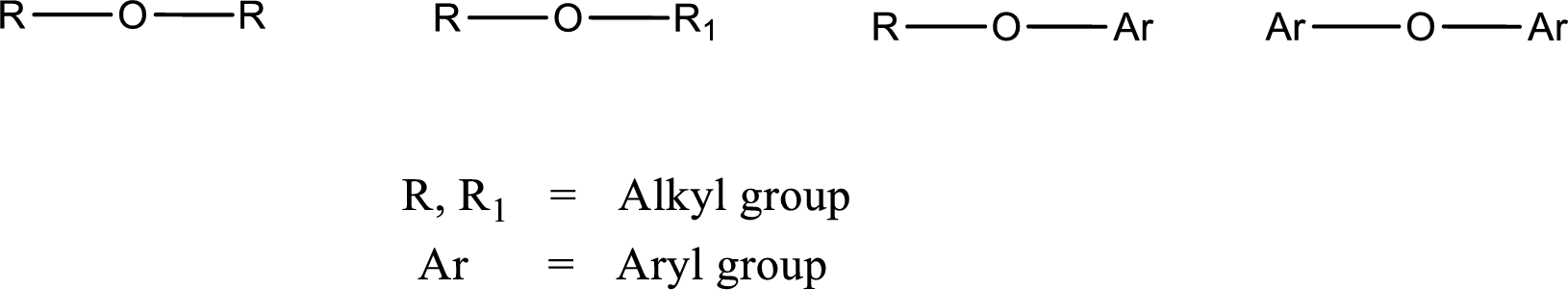
Alcohol is a compound in which a hydrocarbon group is bonded to a hydroxyl group. The generalized formula for alcohol can be given as,
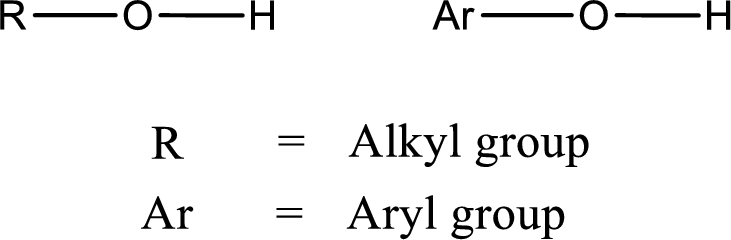
(c)
Interpretation:
The given compound has to be classified as an ether, an alcohol, or neither an ether or alcohol.
Concept Introduction:
Ether is an organic compound. In ether, an oxygen atom is bonded to two carbon atoms by a single bond. The groups that are attached to the carbon atom by a single bond may be alkyl, aryl, or cycloalkyl groups. The functional group of ether is
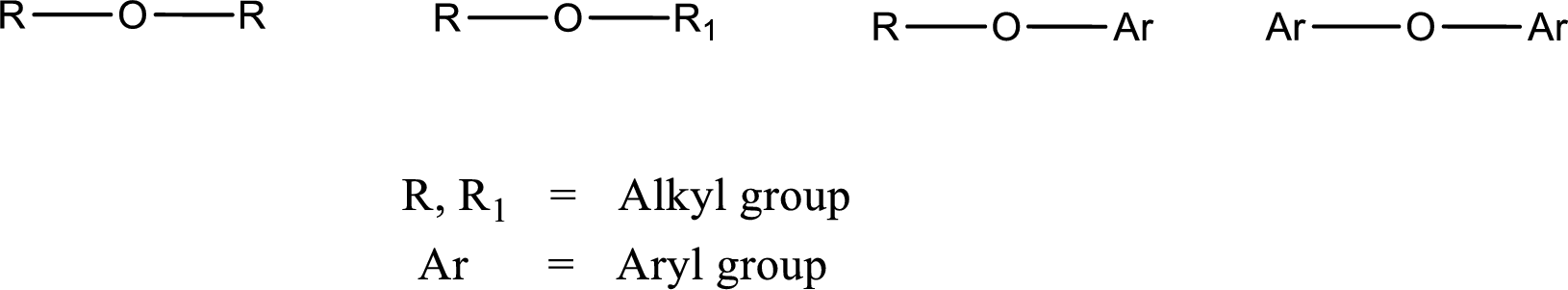
Alcohol is a compound in which a hydrocarbon group is bonded to a hydroxyl group. The generalized formula for alcohol can be given as,
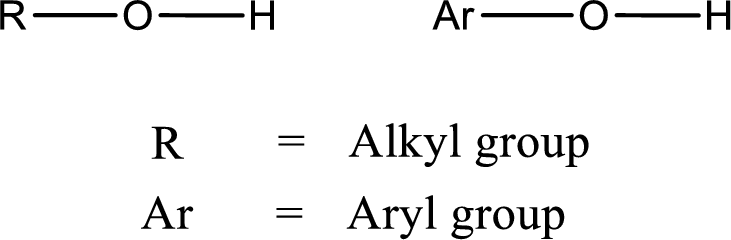
(d)
Interpretation:
The given compound has to be classified as an ether, an alcohol, or neither an ether or alcohol.
Concept Introduction:
Ether is an organic compound. In ether, an oxygen atom is bonded to two carbon atoms by a single bond. The groups that are attached to the carbon atom by a single bond may be alkyl, aryl, or cycloalkyl groups. The functional group of ether is
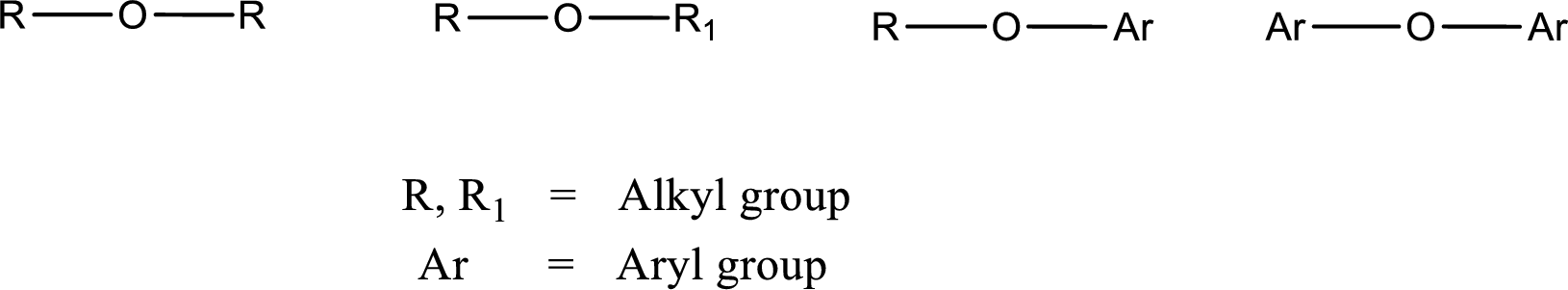
Alcohol is a compound in which a hydrocarbon group is bonded to a hydroxyl group. The generalized formula for alcohol can be given as,
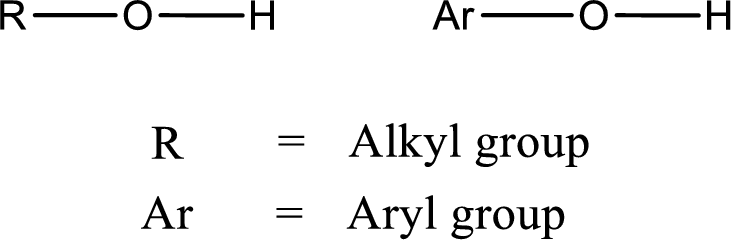
Trending nowThis is a popular solution!

Chapter 14 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Draw the alcohol used to produce each of the following ketones. Include all hydrogen atoms.arrow_forwardOne of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride?arrow_forwardDraw the skeletal structure of the alkene that is needed as a starting material to prepare each of the following alcohols. Part 1 of 2 CH3CH2CH2CH2OH Click and drag to start drawing a structure. ☑ :☐arrow_forward
- Consider the following acids and their ionization constant, determine which conjugate base is HCOOH Ka = 1.7 x 10-4 (b) HCN Ka = 4.9 x 10-10arrow_forwardDraw the skeletal structure for the organic product(s) formed when the following alcohol is dehydrated with H2SO4. OH Click and drag to start drawing a structure. ☐ :arrow_forwardRank the following three compounds in order of decreasing basicity. NH2 NH₂ NH2 NO2 I II III A) II > I > III B) III II I C) III > I > II D) I > II > IIIarrow_forward
- (a) (S)-2-chlorobutane, draw a three-dimensional representation.arrow_forwardللتدة Consider the following compounds. O 2 3 Use the numbers shown to answer the following questions. If none of the structures is applicable enter the word none. An anhydride is represented by the structure numbered O O HC 1 CH3 HC 3 CH3arrow_forwardDefine sterols.arrow_forward
- Draw the structure of the wax composed of stearic acid and a straight-chained alcohol with 30 carbon atoms.arrow_forwardDraw a Lewis structure for hydroxylamine, NH2OH.arrow_forwardWrite the chemical formula for the conjugate base of each of the following acids: Carbonic Acid (H2CO3)___________________ Formic Acid (CH2O2)_____________________arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





