
(a)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
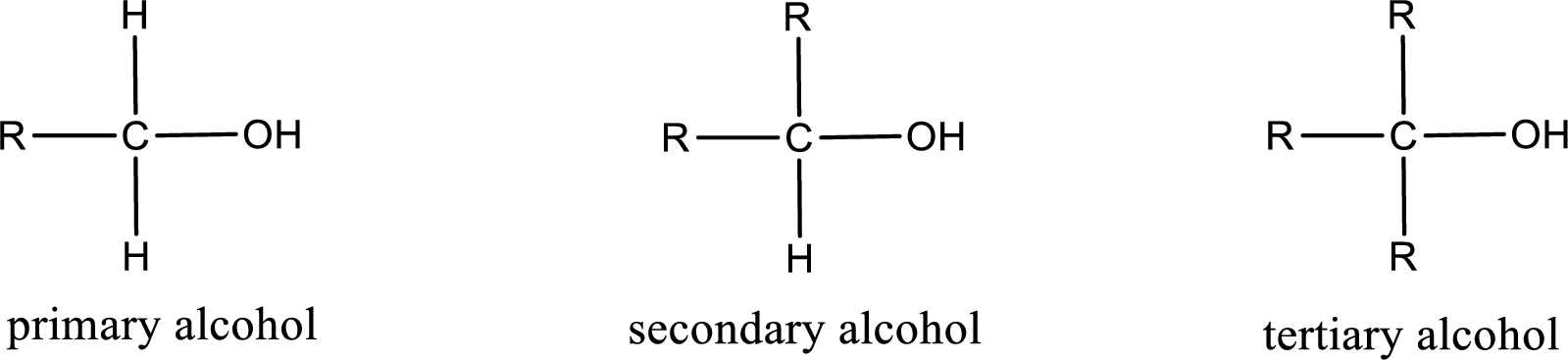
Primary is denoted as
(b)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
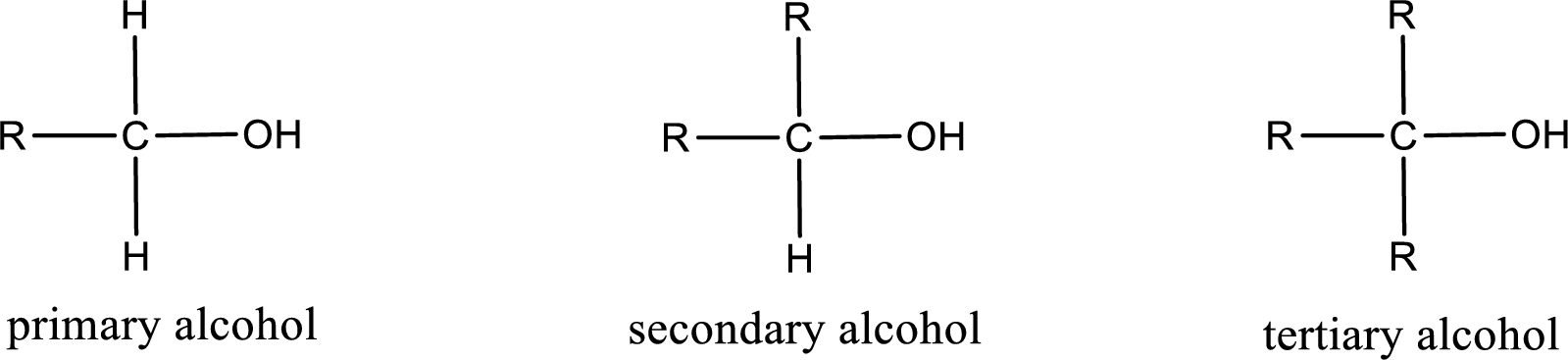
Primary is denoted as
(c)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
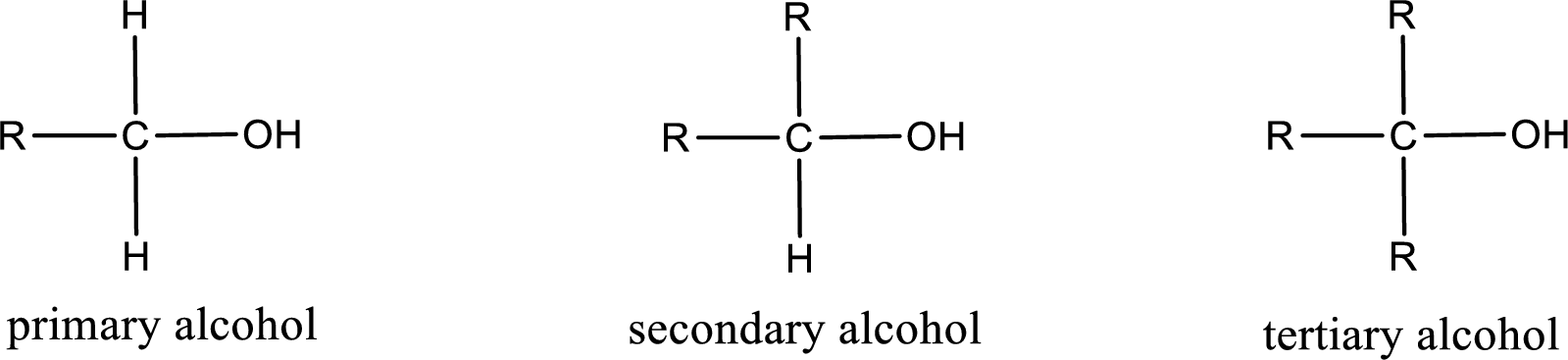
Primary is denoted as
(d)
Interpretation:
The given alcohol is classified as what type of alcohol has to be identified.
Concept Introduction:
Alcohols are classified as primary, secondary, or tertiary depending upon the number of carbon atoms that is bonded to the carbon atom bearing hydroxyl group.
Primary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to only one other carbon atom.
Secondary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to two other carbon atoms.
Tertiary alcohol is the one in which the carbon atom bearing the hydroxyl group is bonded to three other carbon atoms.
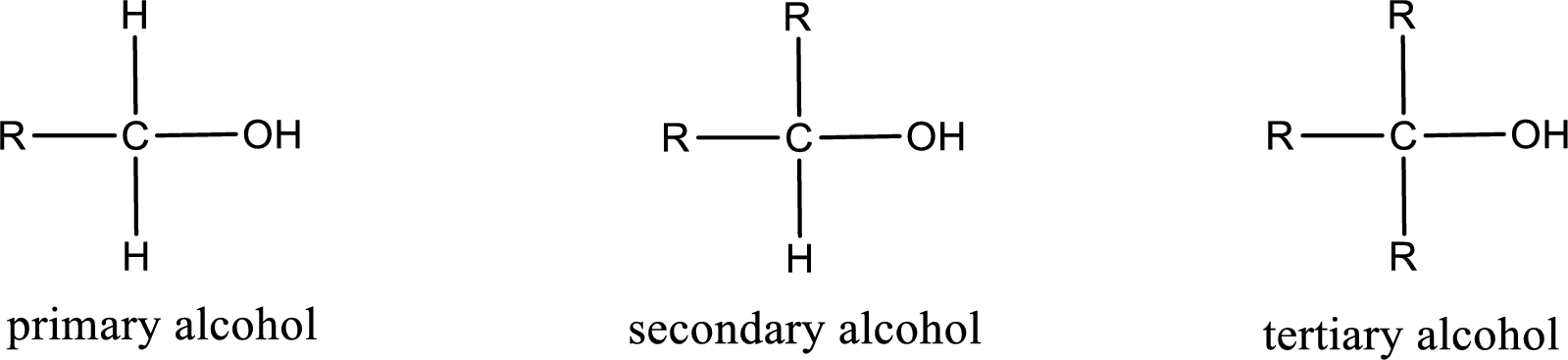
Primary is denoted as
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Classify each of the following alcohols as a primary, secondary, or tertiary alcohol. a. 1-pentanol b. 2-pentanol c. 2-methyl-1-pentanol d. 2-methyl-2-pentanolarrow_forwardClassify the following alcohols as primary, secondary, or tertiary: a. b.CH3CH2CH2CH2OH c.arrow_forwardDefine the Properties of Alcohols ?arrow_forward
- Describe chemical properties of alcohols (intermolecular dehydration, intramolecular dehydration).arrow_forwardAs stated in Section 11-9, the wax found in apple skins is an unbranched alkane with the molecular formula C^H^. Explain how the presence of this alkane in apple skins prevents the loss of moisture from within the apple.arrow_forwardAssign an IUPAC name to each of the compounds in Problem 12-63. a. b. c. d.arrow_forward
- Alcoholic beverages contain: a. wood alcohol. b. isopropyl alcohol. c. glyceryl alcohol. d. ethyl alcohol.arrow_forwardDetermine the maximum number of hydrogen bonds that can form between an ethanol molecule and a. other ethanol molecules b. water molecules c. methanol molecules d. 1-propanol moleculesarrow_forwardGive the IUPAC name of the alcohol that fits each of the following descriptions. a. Moistening agent in many cosmetics b. Major ingredient in environmentally friendly antifreeze formulations c. Industrially produced from CO and H2 d. Often produced via a fermentation processarrow_forward
- Which is NOT a physical property of alcohols or phenols? O Phenols are generally only slightly soluble in water. O The hydroxyl group of an alcohol is nonpolar. The solubilities of normal primary alcohols in water decrease with increasing molecular weight. Boiling points of normal primary alcohols increase with increasing molecular weight.arrow_forwardWhich of the following is incorrect with regards the molecule shown below? OH CH3 CH₂CH--CCH₂ CH3 O it is a secondary alcohol its IUPAC name is 2,2-dimethylbutan-3-ol it is fully saturated (has the max number of H atoms) its IUPAC name is 4,4-dimethylbutan-2-olarrow_forwardClassify and list each oxygen atom in this molecule according to the functional group to which it belongs. if an oxygen is a part of an alcohol classify the alcohol as primary secondary or tertiary.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





