
Concept explainers
Interpretation:
The energies of rotation for ammonia,
Concept introduction:
Atoms of a molecule rotate in space about its moment of inertia. The rotational quantum number is represented by the symbol
Answer to Problem 14.98E
The energies of rotation for ammonia,
| 1 | -1 | 3753.185 |
| 1 | 0 | 1264.06 |
| 1 | 1 | 3753.185 |
| 2 | -2 | 13748.68 |
| 2 | -1 | 6281.305 |
| 2 | 0 | 3792.18 |
| 2 | 1 | 6281.305 |
| 2 | 2 | 13748.68 |
| 3 | -3 | 29986.49 |
| 3 | -2 | 17540.86 |
| 3 | -1 | 10073.49 |
| 3 | 0 | 7584.36 |
| 3 | 1 | 10073.49 |
| 3 | 2 | 17540.86 |
| 3 | 3 | 29986.49 |
| 4 | -4 | 52466.6 |
| 4 | -3 | 35042.73 |
| 4 | -2 | 22597.1 |
| 4 | -1 | 15129.73 |
| 4 | 0 | 12640.6 |
| 4 | 1 | 15129.73 |
| 4 | 2 | 22597.1 |
| 4 | 3 | 52466.6 |
| 4 | 4 | 52466.6 |
| 5 | -5 | 81189.03 |
| 5 | -4 | 58786.9 |
| 5 | -3 | 41363.03 |
| 5 | -2 | 28917.4 |
| 5 | -1 | 21450.03 |
| 5 | 0 | 18960.9 |
| 5 | 1 | 21450.03 |
| 5 | 2 | 28917.4 |
| 5 | 3 | 41363.03 |
| 5 | 4 | 58786.9 |
| 5 | 5 | 81189.03 |
| 6 | -6 | 116153.8 |
| 6 | -5 | 88773.39 |
| 6 | -4 | 66371.26 |
| 6 | -3 | 48947.39 |
| 6 | -2 | 36501.76 |
| 6 | -1 | 29034.39 |
| 6 | 0 | 26545.26 |
| 6 | 1 | 29034.39 |
| 6 | 2 | 36501.76 |
| 6 | 3 | 48947.39 |
| 6 | 4 | 66371.26 |
| 6 | 5 | 88773.39 |
| 6 | 6 | 116153.8 |
| 7 | -7 | 157360.8 |
| 7 | -6 | 125002.2 |
| 7 | -5 | 97621.81 |
| 7 | -4 | 75219.68 |
| 7 | -3 | 57795.81 |
| 7 | -2 | 45350.18 |
| 7 | -1 | 37882.81 |
| 7 | 0 | 35393.68 |
| 7 | 1 | 37882.81 |
| 7 | 2 | 45350.18 |
| 7 | 3 | 57795.81 |
| 7 | 4 | 75219.68 |
| 7 | 5 | 97621.81 |
| 7 | 6 | 125002.2 |
| 7 | 7 | 157360.8 |
| 8 | -8 | 204810.2 |
| 8 | -7 | 167473.3 |
| 8 | -6 | 135114.7 |
| 8 | -5 | 107734.3 |
| 8 | -4 | 85332.16 |
| 8 | -3 | 67908.29 |
| 8 | -2 | 55462.66 |
| 8 | -1 | 47995.29 |
| 8 | 0 | 45506.16 |
| 8 | 1 | 47995.29 |
| 8 | 2 | 55462.66 |
| 8 | 3 | 67908.29 |
| 8 | 4 | 85332.16 |
| 8 | 5 | 107734.3 |
| 8 | 6 | 135114.7 |
| 8 | 7 | 167473.3 |
| 8 | 8 | 204810.2 |
| 9 | -9 | 258501.8 |
| 9 | -8 | 216186.7 |
| 9 | -7 | 178849.8 |
| 9 | -6 | 146491.2 |
| 9 | -5 | 119110.8 |
| 9 | -4 | 96708.7 |
| 9 | -3 | 79284.83 |
| 9 | -2 | 66839.2 |
| 9 | -1 | 59371.83 |
| 9 | 0 | 56882.7 |
| 9 | 1 | 59371.83 |
| 9 | 2 | 66839.2 |
| 9 | 3 | 79284.83 |
| 9 | 4 | 96708.7 |
| 9 | 5 | 119110.8 |
| 9 | 6 | 146491.2 |
| 9 | 7 | 178849.8 |
| 9 | 8 | 216186.7 |
| 9 | 9 | 258501.8 |
| 10 | -10 | 318435.8 |
| 10 | -9 | 271142.4 |
| 10 | -8 | 228827.3 |
| 10 | -7 | 191490.4 |
| 10 | -6 | 159131.8 |
| 10 | -5 | 131751.4 |
| 10 | -4 | 109349.3 |
| 10 | -3 | 91925.43 |
| 10 | -2 | 79479.8 |
| 10 | -1 | 72012.43 |
| 10 | 0 | 69523.3 |
| 10 | 1 | 72012.43 |
| 10 | 2 | 79479.8 |
| 10 | 3 | 91925.43 |
| 10 | 4 | 109349.3 |
| 10 | 5 | 131751.4 |
| 10 | 6 | 159131.8 |
| 10 | 7 | 191490.4 |
| 10 | 8 | 228827.3 |
| 10 | 9 | 271142.4 |
| 10 | 10 | 318435.8 |
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
For the rotational quantum number
The energy level diagram for all the rotational levels is shown below.
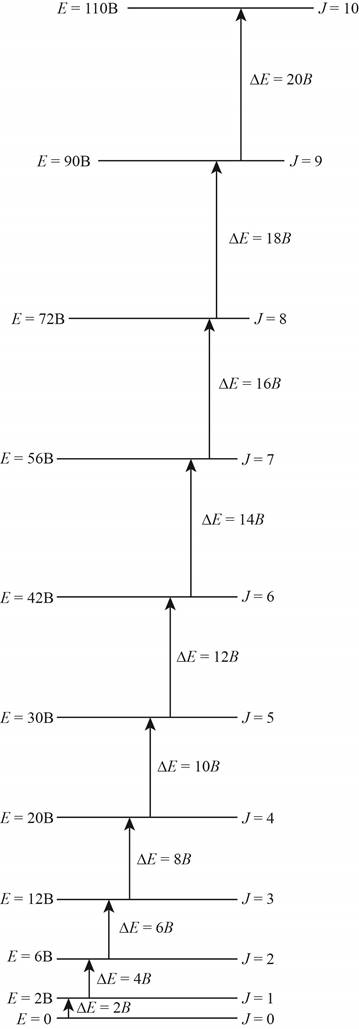
Explanation of Solution
The formula to energy of rotation (
Where,
•
•
The formula for
The formula for
Where,
•
•
The value of
Substitute the value of
The value of
Substitute the value of
The value of
The degeneracy is calculated by the formula given below.
For the rotational quantum number
The value of
The value of
Substitute the value of
Therefore, the degeneracy is
Substitute the value of
Similarly the value of
| 1 | -1 | 3753.185 |
| 1 | 0 | 1264.06 |
| 1 | 1 | 3753.185 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 2 | -2 | 13748.68 |
| 2 | -1 | 6281.305 |
| 2 | 0 | 3792.18 |
| 2 | 1 | 6281.305 |
| 2 | 2 | 13748.68 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 3 | -3 | 29986.49 |
| 3 | -2 | 17540.86 |
| 3 | -1 | 10073.49 |
| 3 | 0 | 7584.36 |
| 3 | 1 | 10073.49 |
| 3 | 2 | 17540.86 |
| 3 | 3 | 29986.49 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 4 | -4 | 52466.6 |
| 4 | -3 | 35042.73 |
| 4 | -2 | 22597.1 |
| 4 | -1 | 15129.73 |
| 4 | 0 | 12640.6 |
| 4 | 1 | 15129.73 |
| 4 | 2 | 22597.1 |
| 4 | 3 | 52466.6 |
| 4 | 4 | 52466.6 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 5 | -5 | 81189.03 |
| 5 | -4 | 58786.9 |
| 5 | -3 | 41363.03 |
| 5 | -2 | 28917.4 |
| 5 | -1 | 21450.03 |
| 5 | 0 | 18960.9 |
| 5 | 1 | 21450.03 |
| 5 | 2 | 28917.4 |
| 5 | 3 | 41363.03 |
| 5 | 4 | 58786.9 |
| 5 | 5 | 81189.03 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 6 | -6 | 116153.8 |
| 6 | -5 | 88773.39 |
| 6 | -4 | 66371.26 |
| 6 | -3 | 48947.39 |
| 6 | -2 | 36501.76 |
| 6 | -1 | 29034.39 |
| 6 | 0 | 26545.26 |
| 6 | 1 | 29034.39 |
| 6 | 2 | 36501.76 |
| 6 | 3 | 48947.39 |
| 6 | 4 | 66371.26 |
| 6 | 5 | 88773.39 |
| 6 | 6 | 116153.8 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 7 | -7 | 157360.8 |
| 7 | -6 | 125002.2 |
| 7 | -5 | 97621.81 |
| 7 | -4 | 75219.68 |
| 7 | -3 | 57795.81 |
| 7 | -2 | 45350.18 |
| 7 | -1 | 37882.81 |
| 7 | 0 | 35393.68 |
| 7 | 1 | 37882.81 |
| 7 | 2 | 45350.18 |
| 7 | 3 | 57795.81 |
| 7 | 4 | 75219.68 |
| 7 | 5 | 97621.81 |
| 7 | 6 | 125002.2 |
| 7 | 7 | 157360.8 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 8 | -8 | 204810.2 |
| 8 | -7 | 167473.3 |
| 8 | -6 | 135114.7 |
| 8 | -5 | 107734.3 |
| 8 | -4 | 85332.16 |
| 8 | -3 | 67908.29 |
| 8 | -2 | 55462.66 |
| 8 | -1 | 47995.29 |
| 8 | 0 | 45506.16 |
| 8 | 1 | 47995.29 |
| 8 | 2 | 55462.66 |
| 8 | 3 | 67908.29 |
| 8 | 4 | 85332.16 |
| 8 | 5 | 107734.3 |
| 8 | 6 | 135114.7 |
| 8 | 7 | 167473.3 |
| 8 | 8 | 204810.2 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 9 | -9 | 258501.8 |
| 9 | -8 | 216186.7 |
| 9 | -7 | 178849.8 |
| 9 | -6 | 146491.2 |
| 9 | -5 | 119110.8 |
| 9 | -4 | 96708.7 |
| 9 | -3 | 79284.83 |
| 9 | -2 | 66839.2 |
| 9 | -1 | 59371.83 |
| 9 | 0 | 56882.7 |
| 9 | 1 | 59371.83 |
| 9 | 2 | 66839.2 |
| 9 | 3 | 79284.83 |
| 9 | 4 | 96708.7 |
| 9 | 5 | 119110.8 |
| 9 | 6 | 146491.2 |
| 9 | 7 | 178849.8 |
| 9 | 8 | 216186.7 |
| 9 | 9 | 258501.8 |
For the rotational quantum number
Substitute the value of
Therefore, the degeneracy is
Similarly the value of
| 10 | -10 | 318435.8 |
| 10 | -9 | 271142.4 |
| 10 | -8 | 228827.3 |
| 10 | -7 | 191490.4 |
| 10 | -6 | 159131.8 |
| 10 | -5 | 131751.4 |
| 10 | -4 | 109349.3 |
| 10 | -3 | 91925.43 |
| 10 | -2 | 79479.8 |
| 10 | -1 | 72012.43 |
| 10 | 0 | 69523.3 |
| 10 | 1 | 72012.43 |
| 10 | 2 | 79479.8 |
| 10 | 3 | 91925.43 |
| 10 | 4 | 109349.3 |
| 10 | 5 | 131751.4 |
| 10 | 6 | 159131.8 |
| 10 | 7 | 191490.4 |
| 10 | 8 | 228827.3 |
| 10 | 9 | 271142.4 |
| 10 | 10 | 318435.8 |
The energy level diagram for all the rotational levels is shown below.
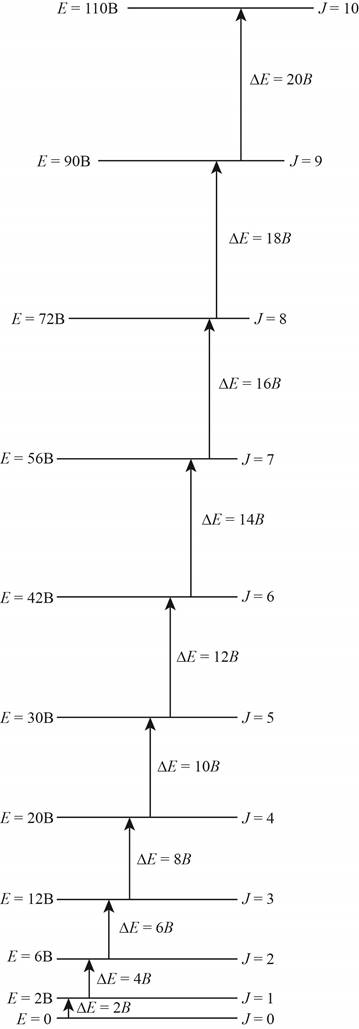
Figure 1
The energies of rotation for ammonia,
Want to see more full solutions like this?
Chapter 14 Solutions
Bundle: Physical Chemistry, 2nd + Student Solutions Manual
- A space probe was designed to see 13CO in the atmosphere of Saturn by looking for lines in its rotational spectrum. If the bond length of CO is 112.8 pm, at what wave number do the first three rotational transitions appear?arrow_forwardEmission of microwave radiation from the J = 10 transition of a molecule has been detected at 88.63 GHz from a region of interstellar space in which there is evidence of thermal equilibrium and a temperature of around 50 K. Estimate the frequency and relative intensity of the J = 2 → 1 transition of the same molecule.arrow_forward8C.4 (a) the moment of inertia of a CH4 molecule is 5.27 x 10^-47 kg m^2. What is the minimum energy needed to start it rotating? 8C.5 (a) use the data in 8C.4 (a) to calculate the energy needed excite a CH4 molecule from a state with l=1 to a state with l=2arrow_forward
- 559.7 cm¹, and a rotational constant, B = 0.244 cm ¹¹. 18. Chlorine has a vibrational constant, U = From this information, determine (a) the force constant and (b) the equilibrium bond length.arrow_forwardExplain the importance of the quantization of vibrational, rotational, and translational energy as it relates to the behavior of atoms and molecules.arrow_forwardAn HCl molecule initially in the (v, J) = (0, 2) quantum state can absorb infrared photons. What are the two most likely final quantum states after absorbing an infrared photon? Calculate the energy of the infrared photonabsorbed for each of these transitions. Can it also absorb a microwave photon? If yes, calculate that photon energy also. The rotational constant of HCl is 10 cm-1 and its vibrational frequency is 2990 cm-1.arrow_forward
- The rotational spectrum of CH* has been observed in the planetary nebula NGC 7027." The spectrum shows a series of lines separated by 29.64 cm1. Calculate the bond length of CH*. Take the reduced mass of CH* to be 0.9299 g/mol. Give your answer in angstroms with two decimal places. Answer:arrow_forwardThe moment of inertia of CH4 can be calculated from the expression I=8/3 mHR2 where R is the C-H bond length (109 angstrom or 109 x 1012 m). a. What is the lowest possible rotational energy of the CH4 molecule and what is the value of quantum number l associated with that rotational energy? b. Calculate the rotational energy of the molecule in the first excited state (when quantum number l = 1). c. Determine the degeneracy of the first excited state. Explain what is meant by rotational energy degeneracy.arrow_forwardWhat is the speed of a photoelectron ejected from an orbital of ionization energy 12.0 eV by a photon of radiation of wavelength 100 nm?arrow_forward
- When β-carotene is oxidized in vivo, it breaks in half and forms two molecules of retinal (vitamin A), which is a precursor to the pigment in the retina responsible for vision. The conjugated system of retinal consists of 11 C atoms and 1 O atom. In the ground state of retinal, each level up to n = 6 is occupied by two electrons. Assuming an average internuclear distance of 140 pm, calculate (a) the separation in energy between the ground and first exciteted state in which one electron occupies the state with n = 7, and (b) the frequency of radiation required to produce a transition between these two states. From your results, correct the following sentence (from the options in brackets). The absorption spectrum of a linear polyene shifts to (higher/lower) frequency as the number of conjugated atoms (increases/decreases).arrow_forwardA molecule can have various types of energies (translational, rotational, vibrational, and electronic), the sum of which is the molecule's total energy. E trans = (n +n + n²) Erot = J (J + 1) h² 87²1 Evib = (U+ 1 ) h hv h² 8mV (2/3) In the equations, nx, ny, nz, J, and u are quantum numbers, h is Planck's constant, m is the mass of the molecule, V is the volume of the container, I is the moment of inertia of the molecule, and v is the fundamental vibration frequency. For carbon monoxide, CO, the moment of inertia is I = 1.45 x 10-46 kg-m², and the fundamental vibration frequency is v = 2130 cm-¹. Let V = 12.5 L, and let all the quantum numbers be equal to 1. Calculate the translational, rotational, and vibrational energies per mole of CO for these conditions.arrow_forwardIn the methane molecule, CH4, each hydrogen atom is at the corner of a regular tetrahedron with the carbon atom at the center. If one of the C-H is in the direction of A= î +7 + k and an adjacent C-H bond is at the direction B=î-7-R. results to an angular bond of approximately 109° for a static frozen molecule. However, the molecule we can encounter everyday continuously vibrates and interact with the surrounding causing its bond vector to vary slightly. According to a new spectroscopy analysis, the adjacent bond vectors was found to be A = 1.08i + 0.89j + 1.08k B = 0.85i + -0.93j + -0.94k What is the angle (in degrees) between the bonds based on this new data? Note: Only 1% of error is permitted for the correct answer. Iarrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,

