
A volume of air (assumed to be an ideal gas) is first cooled without charging its volume and then expanded without changing its pressure, as shown by the path abc in Figure 15.35. (a) How does the final temperature of the gas compare with its initial temperature? (b) How much heat does the air exchange with its surroundings during the process abc? Does the air absorb heat or release heat during this process? Explain. (c) If the air instead expands from state a to state c by the straight-line path shown, how much heat does it exchange with its surroundings?
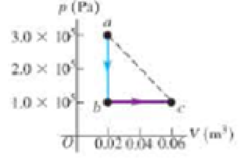
Figure 15.35
Problem 52.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
College Physics (10th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
The Cosmic Perspective Fundamentals (2nd Edition)
An Introduction to Thermal Physics
Lecture- Tutorials for Introductory Astronomy
Physics: Principles with Applications
- An aluminum rod 0.500 m in length and with a cross-sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 300 K. (a) If one-half of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool. (b) If the circular surface of the upper end of the rod is maintained at 300 K, what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 W/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forwardAssume Lake Erie contains 4.80 ✕ 1011 m3 of water, and assume the water's density is that of water at 20°C and 1 atm. (a) How much energy (in J) is required to raise the temperature of that volume of water from 15.8°C to 23.4°C? ?J (b) How many years would it take to supply this amount of energy by using a power of 1,400 MW generated by an electric power plant? ?yrarrow_forwardCalculate how much heat in J has to be added to a sample of an ideal monoatomic gas at a pressure of 2.1x105 N/m2 and a temperature of 292 K so it undergoes a quasi-static isobaric expansion from 2.0x103 cm3 to 4.0x103 cm3arrow_forward
- Jill takes in 0.0140 mol of air in a single breath. The air is taken in at 20.0°C and exhaled at 35.0°C. Her respiration rate is (2.00x10^1) breaths per minute. At what average rate does heat leave her body due to the temperature increase of the air? Provide your answer to three significant figures. HINT: Use the molar specific heat at constant volume to find the heat loss, where Cv = 5R/2 (for an ideal diatomic gas).arrow_forwardConsider an oil-to-oil double-pipe heat exchanger whose flow arrangement is not known. The temperature measurements indicate that the cold oil enters at 20°C and leaves at 55°C, while the hot oil enters at 80°C and leaves at 45°C. Do you think this is a parallel-flow or counter-flow heat exchanger? Why? Assuming the mass flow rates of both fluids to be identical, determine the effectiveness of this heat exchanger.arrow_forwardIf you have 3 moles of a monoatomic ideal gas, how much heat is required to raise the temperature of this gas from 261.8K to 284.8K if the volume of the gas remains constant during the heating?arrow_forward
- Lake Erie contains roughly 4.00 ✕ 1011 m3 of water. (a) How much energy is required to raise the temperature of that volume of water from 14.8°C to 23.6°C? (Assume the density of this water to be equal to that of water at 20°C and 1 atm.) J(b) How many years would it take to supply this amount of energy by using the 1,400-MW exhaust energy of an electric power plant?arrow_forwardIf you have 4 moles of a diatomic ideal gas (such as oxygen or nitrogen), how much heat is required to raise the temperature of this gas from 269.5K to 288.3K if the volume of the gas remains constant during the heating?arrow_forwardA 0.617 mol sample of Xe(g) initially at 298 K and 1.00 atm is held at constant volume while enough heat is applied to raise the temperature of the gas by 14.9 K. Assuming ideal gas behavior, calculate the amount of heat (?)in joules required to affect this temperature change and the total change in internal energy, Δ?. Note that some books use Δ? as the symbol for internal energy instead of Δ?. Type of gas Molar heat capacity at constant volume (??,?) atoms (3/2)? linear molecules (5/2)? nonlinear molecules 3? where ? is the ideal gas constantarrow_forward
- A sample of gas in a cylinder with a piston is in thermal contact with a heat reservoir at a temperature of 353 K. While keeping this gas at this constant temperature and at a constant pressure of 1.10 105 N/m2, we permit the gas to expand by 1.60 10-5 m3. How much heat does the gas absorb during this process?arrow_forwardHow many molecules are present in a sample of an ideal gas that occupies a volume of 1.70 cm3, is at a temperature of 20°C, and is at atmospheric pressure? How many molecules of the gas are present if the volume and temperature are the same as in part (a), but the pressure is now 2.80 ✕ 10−11 Pa (an extremely good vacuum)?arrow_forwardA thirsty nurse cools a 2.30 LL bottle of a soft drink (mostly water) by pouring it into a large aluminum mug of mass 0.257 kgkg and adding 0.123 kgkg of ice initially at -14.0 ∘C∘C. If the soft drink and mug are initially at 20.9 ∘C∘C, what is the final temperature of the system, assuming no heat losses?arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

