
(a)
Interpretation: The maximum density of water needs to be determined.
Concept Introduction: Density is defined as the mass of a substance occupied per unit of volume. It changes with temperature.
(a)
Explanation of Solution
The given graph shows the density of water over the temperature range
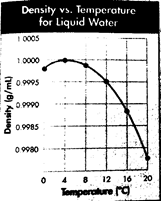
From the graph, it can be seen that the maximum density of water is 1.0000 g/mL. This is the point at the peak of the curve obtained when density and temperature are plotted.
Therefore, the maximum density of water is 1.0000 g/mL.
(b)
Interpretation: The temperature at which the maximum density of water occurs needs to be determined.
Concept Introduction: Density is defined as the mass of a substance occupied per unit of volume. It changes with temperature.
(b)
Explanation of Solution
The given graph shows the density of water over the temperature range
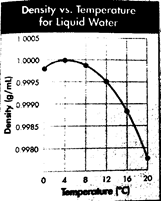
From the graph, it can be seen that the maximum density of water is 1.0000 g/mL. This is the point at the peak of the curve obtained when density and temperature are plotted. Now, the point meets the x-axis (where the temperature is plotted) at
Therefore, the temperature at which the maximum density of water occurs is
(c)
Interpretation: Whether it is meaningful to expand the smooth curve if the graph to the left to temperatures below
Concept Introduction: The state of water also changes with a change in temperature. At
(c)
Explanation of Solution
The state of water below
The main reason is that liquid water changes its state from liquid to solid (ice) at
Chapter 15 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





