
Concept explainers
PRACTICE PROBLEM 15.8
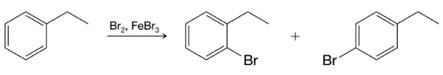
Write resonance structures for the arenium ions formed when ethylbenzene reacts with a
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Chemistry: Structure and Properties (2nd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Introductory Chemistry (5th Edition) (Standalone Book)
- In an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?arrow_forwardWhich of the following synthesis reactions of organohalide occurs via electrophilic addition? (A) reaction of chlorine with propane B reaction of chlorine with propene reaction of thionyl chloride with propan-2-ol (D) all choicesarrow_forward(b) State the reagents needed to convert benzoic acid into the following compounds. (i) C6H§COCI (ii) C,H$CH2OH (iii) C6H$CONHCH3arrow_forward
- Write structural formulas for the cyclohexadienyl cations formed from aniline (C6H5NH2) during(a) Ortho bromination (four resonance structures)(b) Meta bromination (three resonance structures)(c) Para bromination (four resonance structures)arrow_forwardWhich of the following reactions will synthesize phenol from benzene? 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) warm H2SO4 and H2O 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) CuCN; 5) dilute acid and heat 1) Acetyl chloride & AlCl3; 2) bleach 1) Ph-N2+ + KI; 2) BrMgCH=CH2 in ether, followed by H3O+; 3) warm, conc'd KMnO4 1) Cl-CH(CH3)-CH2CH2CH3 + FeBr3; 2) hot, conc'd KMnO4arrow_forward(a) Tsomane and Nyiko were given a task of synthesising methylenecyclohexane 2. After a brief discussion with each other, Tsomane proposed Method A to synthesise 2 from cyclohexanone 1 while Nyiko proposed Method B that started from hydroxymethylcyclohexane 3. Each student believed that their proposed method is better than the other. (Scheme below) (1) 1 Ph THF A Ph Ph B H₂SO4 100 °C 3 OH What is the name of the reaction that is followed by reaction Method A?arrow_forward
- Compound F may be synthesised by the method attached: When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce two different functional groups. Provide both reactions and explain the two different functional groups produced.arrow_forwardPredict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents.(a) ethanoarrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents.(a) HNO3, H2SO4 (b) Br2, FeBr3 (c) CH3CH2COCl, AlCl3(d) isobutylene and HFarrow_forward
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents.(a) HNO3, H2SO4 (b) Br2, FeBr3 (c) CH3CH2COCl, AlCl3arrow_forward(a) Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.(b) How will you bring about the following converstions?(i) Propanone to propane (ii) Benzoyl chloride to benzaldehyde(iii) Ethanal to but-2-enalarrow_forward(d) How would you prepare any one of the following compounds from benzene? More than one step may be involved in each case. (a) (b) OH Br m-Bromo benzoic acid Phenyl acetic acidarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





