
Concept explainers
(a)
Interpretation:
The following pair of molecules should be classified as stereoisomers, constitutional isomers, or identical molecules:

Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms or group of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
(b)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:
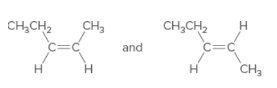
Concept Introduction:
Isomers are the compounds having the same molecular formula but different structures. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have the same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having the same bond connectivity and same molecular formula are known as identical molecules.
(c)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:

Concept Introduction:
Isomers are the compounds having same molecular formula but different structures. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have the same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having the same bond connectivity and same molecular formula are known as identical molecules.
(d)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:

Concept Introduction:
Isomers are the compounds having the same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have the same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having the same bond connectivity and same molecular formula are known as identical molecules.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Which of the following pairs represent structural isomers, and which are simply the same compound? a. and CH3CH2CH2CH3 b. and CH3CH2CH2CH2CH2CH3 c. and CH3CH2CH2CH3 d. andarrow_forwardDraw the four constitutional isomers having molecular formula C4H9Br and indicate how many different kinds of carbon atoms each has.arrow_forwardHow many structural isomers of C4H6 have exactly one ring?arrow_forward
- Organic compounds may have characteristic odors as well as other characteristic physical properties. For example, the distinct odor of the seashore at low tide results in part from the presence of dimethyl sulfide (CH3SCH3), a molecule with a similar structure to dimethyl ether (CH3OCH3). Ethanethiol (CH3CH2SH), also called mercaptan, is an isomer of dimethyl sulfide with a much less pleasant odor.The table lists four related compounds and their enthalpies of vaporization (ΔH°vap) in kJ/mol. Compound ΔH°vap (kJ/mol) CH3OCH3 23 CH3SCH3 28 CH3CH2SH 27.5 CH3CH2OH 42 Rank the following compounds in order of increasing strength of their intermolecular forces, given the ΔH°vap listed for each. Place the compound with the strongest intermolecular forces (IMFs) at the top of the list. (Strongest to weaknest). Why is ΔHºvap for CH3SCH3 greater than ΔHºvap for CH3OCH3? A. CH3OCH3 is more polar. B. CH3SCH3 has stronger dipole–dipole attractions. C. CH3OCH3 can form…arrow_forward2. Determine which of the structures are isomers of each other and which are conformers of each other. c-c-c-C c-c-c- --- C-C-C-C C-C-C '' C-C-C-C سے ہے ہے ہے دل اليد <-٤-٤ C c- ؟-؟ Carrow_forwardFive structural isomers, or constitutional isomers, have the formula C6H14C6H14. Draw the indicated isomers, grouped by number of carbon atoms in the main chain. a) Draw two isomers with 5 carbon atoms in the main chain. Be sure to include all hydrogen atoms. b) Draw two isomers with 4 carbon atoms in the main chain. Be sure to include all hydrogen atoms.arrow_forward
- 1-bromo-3-ethylcyclopentane exhibits geometrical isomerism. Draw structures of the two geometrical isomersarrow_forward2-methylhexane & 3,3-dimethylpentane Are the two compounds Constitutional Isomers or Different Compounds? Draw structures for both compounds.arrow_forwardDraw the indicated number of constitutional isomers for C4H10 (2 isomers) and C5H12 (3 isomers)arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





