
(a)
Interpretation:
The noncarboxyl
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
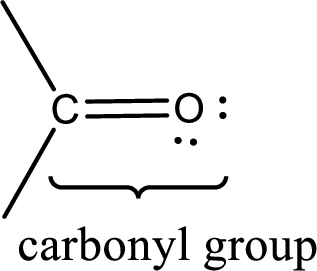
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
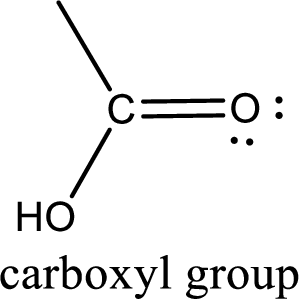
Apart from carboxyl group, there can be other functional group also that is present in the
(b)
Interpretation:
The noncarboxyl functional group that is present in lactic acid has to be given.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
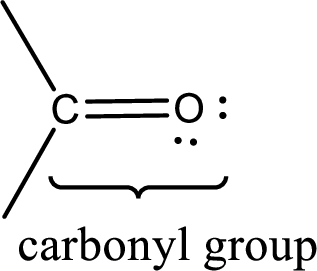
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
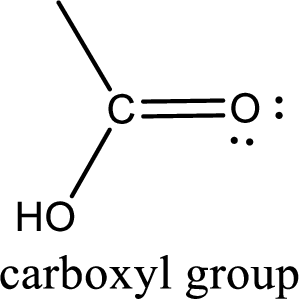
Apart from carboxyl group, there can be other functional group also that is present in the carboxylic acid. They are known as polyfunctional carboxylic acids. These occur naturally also. Some of the important type of polyfunctional carboxylic acids are hydroxy acids, keto acids, and unsaturated acids.
(c)
Interpretation:
The noncarboxyl functional group that is present in maleic acid has to be given.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
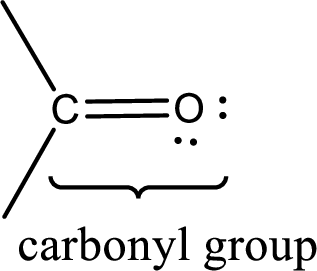
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
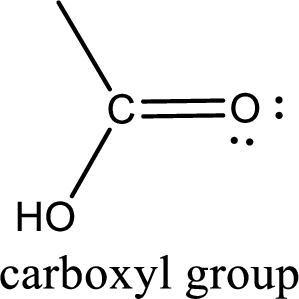
Apart from carboxyl group, there can be other functional group also that is present in the carboxylic acid. They are known as polyfunctional carboxylic acids. These occur naturally also. Some of the important type of polyfunctional carboxylic acids are hydroxy acids, keto acids, and unsaturated acids.
(d)
Interpretation:
The noncarboxyl functional group that is present in glycolic acid has to be given.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
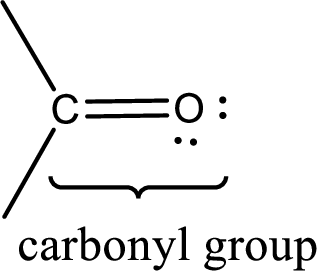
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
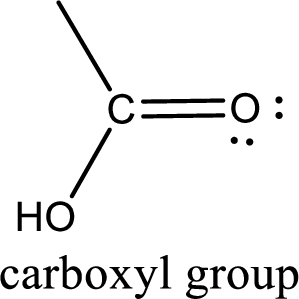
Apart from carboxyl group, there can be other functional group also that is present in the carboxylic acid. They are known as polyfunctional carboxylic acids. These occur naturally also. Some of the important type of polyfunctional carboxylic acids are hydroxy acids, keto acids, and unsaturated acids.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Each of the following acids contains an additional type of functional group besides the carboxyl group. For each acid, specify the non-carboxyl functional group present. 1.Malic acid2. Tartaric acidarrow_forward1. Describe the structure and properties of carboxylic acids and esters. 2. Name at least two (2) common carboxylic acids and esters.arrow_forwardComplete the table provided below for nomenclature of Carboxylic Acid and Derivativesarrow_forward
- Each of the following acids contains an additional type of functional group besides the carboxyl group. For each acid, specify the non-carboxyl functional group present. 12. Glycolic acid13. Fumaric acid14. Pyruvic acid15. Malic acid16. Tartaric acidarrow_forwardwhy n-hexanol has higher boiling point than n-hexane. why acetic acid is more acidic then butanoic acid. why sugar is soluble in water in terms of their molecular interactions.arrow_forwardEach of the following acids contains an additional type of functional group besides the carboxyl group. For each acid, specify the non-carboxyl functional group present. 9. Acrylic acid10. Lactic acid11. Maleic acid12. Glycolic acid13. Fumaric acid14. Pyruvic acid15. Malic acid16. Tartaric acidarrow_forward
- What are the properties of carboxylic acid? What are the steps in the IUPAC and common naming of carboxylic acid?arrow_forwardDefine physical properties of carboxylic acid derivatives ?arrow_forwardAcid Alcohol Odor Structure salicylic acid methanol wintergreen ? anthranilic acid methanol Grape ?arrow_forward
- explain the structures and properties of functional group :carboxylic acid and aminoarrow_forward24.Which type of functional group does not include a carbonyl group in its structure? Multiple Choice aldehyde carboxylic acid ether ketone esterarrow_forwardRefer to the structures of carboxylic acid and derivatives. Which of the following statements is TRUE? Compound A is the most reactive. The second most reactive is compound B Compound D is the least reactive Compound E is the most reactive.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


