
Organic Chemistry - Access (Custom)
4th Edition
ISBN: 9781259355844
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 16.36P
Explain each statement using resonance theory.
a. The indicated
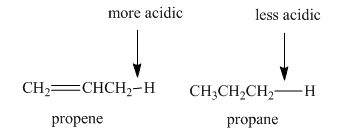
b. The
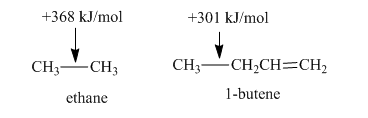
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the hybridized bonds and bond type of the molecules in this reaction?
propan-1-ol → propene + water
C₃H₈O (l) → C3H6 (g) + H2O (l)
1. What is the bond angle in carbon tetrachloride?
2. What is the product of the reaction of pent-2-ene with Cl2?
3.Which statement is correct? *
A. The bond length increases when the difference between the electronegativity of the atoms is higher
B. The bond length increases when the number when there is an increase in pi bonds.
C. Increasing the bond polarity increases the bond length.
D. Sp2 has shorter bond length that sp.
4. How many moles of O2 gas is theoretically needed for 1 mole of hexane?
5. What is the MOST LIKELY product of the reaction of pent-1-ene with HCL?
6. Which one creates a sigma bond? *
A. 2 pz atomic orbitals
B. 2 py atomic orbitals
C. 2 px atomic orbitals
D. none of these
7.How many hydrogen atoms are there in trans-1-bromo-2-methylcyclohexane?
8.
Hybrid orbitals in the triple-bonded carbon of alkynes
Select one:
a. spd
b. sp3
c. sp
d. sp2
Chapter 16 Solutions
Organic Chemistry - Access (Custom)
Ch. 16 - Prob. 16.1PCh. 16 - Prob. 16.2PCh. 16 - Draw a second resonance structure for each...Ch. 16 - Prob. 16.4PCh. 16 - Prob. 16.5PCh. 16 - Prob. 16.6PCh. 16 - Prob. 16.7PCh. 16 - Determine the hybridization of the labeled atom in...Ch. 16 - Problem 16.10 Draw the structure consistent with...Ch. 16 - Problem 16.11 Neuroprotectin D1 (NPD1) is...
Ch. 16 - Problem 16.12 Using hybridization, predict how the...Ch. 16 - Problem 16.13 Use resonance theory to explain why...Ch. 16 - Prob. 16.13PCh. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - Problem 16.17 Draw a stepwise mechanism for the...Ch. 16 - Prob. 16.17PCh. 16 - Problem 16.19 Draw the product formed when each...Ch. 16 - Prob. 16.19PCh. 16 - Prob. 16.20PCh. 16 - Rank the following dienophiles in order of...Ch. 16 - Prob. 16.22PCh. 16 - Prob. 16.23PCh. 16 - Prob. 16.24PCh. 16 - Prob. 16.25PCh. 16 - Problem 16.27 Which compound in each pair absorbs...Ch. 16 - Prob. 16.27PCh. 16 - 16.29 Name each diene and state whether the...Ch. 16 - Prob. 16.29PCh. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Prob. 16.32PCh. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - 16.35 Explain why the cyclopentadienide anion A...Ch. 16 - Explain each statement using resonance theory. a....Ch. 16 - 16.37 Draw the structure of each compound.
a. in...Ch. 16 - Draw and name all dienes of molecular formula...Ch. 16 - Prob. 16.39PCh. 16 - 16.39 Label each pair of compounds as...Ch. 16 - Prob. 16.41PCh. 16 - 16.41 Draw the products formed when each compound...Ch. 16 - Prob. 16.43PCh. 16 - 16.43 Treatment of alkenes A and B with gives the...Ch. 16 - 16.44 Draw a stepwise mechanism for the following...Ch. 16 - Prob. 16.46PCh. 16 - 16.46 Explain, with reference to the mechanism,...Ch. 16 - Prob. 16.48PCh. 16 - Prob. 16.49PCh. 16 - Prob. 16.50PCh. 16 - Prob. 16.51PCh. 16 - Prob. 16.52PCh. 16 - Prob. 16.53PCh. 16 - 16.53 Diels–Alder reaction of a monosubstituted...Ch. 16 - Prob. 16.55PCh. 16 - Prob. 16.56PCh. 16 - 16.55 Devise a stepwise synthesis of each compound...Ch. 16 - Prob. 16.58PCh. 16 - 16.57 A transannular Diels–Alder reaction is an...Ch. 16 - Draw a stepwise mechanism for the following...Ch. 16 - Draw the products of each reaction. Indicate the...Ch. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - 16.65 The treatment of isoprene with one...Ch. 16 - 16.66 The treatment of with forms B (molecular...Ch. 16 - Rank the following compounds in the order of...Ch. 16 - Prob. 16.68PCh. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Prob. 16.73PCh. 16 - Prob. 16.74P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Choose one answer for the following. Refer to the diagram below. 1. Hybridization of atom labelled A (sp, sp2, or sp3) 2. Hybridization of atom labelled B (sp, sp2, or sp3) 3. Hybridization of atom labelled C (sp, sp2, sp3) 4.Class compound for functional group D (phenols, aryl halide, alcohols, aromatics, or carboxylic acids) 5. Class compound for functional group E (phenols, aryl halide, alcohols, aromatics, or carboxylic acids)arrow_forwardb. The C-C bond angles for the ring in M2 is: 60º 109.5º 120º 180 c. What is the oxidation level of M1? Alkane Alcohol Carboxylic Acid Carbon Dioxidearrow_forwardA pair of electrons(s) in the s bond spreads out into empty p orbital, stabilizing electron-deficient carbon atom, the process is known as ________. a) Inductive effect, b) Resonance, c) Hyperconjugation. The carbon atom in dibromocarbene has only ______ electrons in its valence shell with _____ hybridized and ________ geometry. a) Four, sp3, trigonal, b) Five, sp, planar, c) Six, sp2, trigonal, d) Five, sp3, planar.arrow_forward
- Of the benzene molecule it is true that: a. c-c bonds have different bond lengths since they are single and double bonds. b.carries out aromatic electrophilic addition reactions c. the actual structure is a resonance hybrid d.it can be correctly represented with an equilibrium between two resonant structures.arrow_forwardWhy is benzene less reactive than hexane in terms of stability of free radicals?arrow_forward1.1 The density of a gas was measured at 1 50 atm and 27 degree celsuis and found to be 1 95 g/L. Calculate the molar mass of the gas. 1.2 Draw and name three structural isomers of octane (C8H18) 1.3 State Markonikov's rule 1.4 The base Ca(OH)2 is almost insoluble in water , only o 25 g can be dissolved in 0.5 L of water at 25 degree celsuis. If the dissolved substance is completely dissociated into constuent ions , what is the pH of the solution?arrow_forward
- Using the trans effect and the starting reagent [PtCl4]2- indicate the sequence of addition of NH3 and CN- for the preparation of cis-[PtNH3(CN)Cl2] and trans-[PtNH3(CN)Cl2] isomers.arrow_forwardBy giving an example for each of the molecules of Alkyl halide, Aldehyde, Ketone, Carboxylic Acid, Ester, Anhydride, Amit, you can also identify the bond types (sigma -pi) and hybridization typesarrow_forwardExplain why acetic acid, CH3CO2H, Contains two distinct types of Carbon oxygen Bonds, Whereas the acetate ion, formed by loss of hydrogen ion from acetic acid, Only contains one type of carbon oxygen bondarrow_forward
- Define Radicals, Carbocations, and Carbanions ?arrow_forwardORGANIC CHEMISTRY What is the best possible structure for 3-hexyne?arrow_forward1. The hybridization of the functional carbons in alkynes is sp2 while those of alkenes is sp. True False 2. Internal alkynes will produce only one type of product in electrophilic addition. True False 3. The reaction common to both alkenes and alkynes that results in the formation of a new bond between the functional carbon and the reagent is electrophilic addition radical addition electrophilic substitution nucleophilic additionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY