
Interpretation:
Reason for the difference in the given boiling points of
Concept introduction:
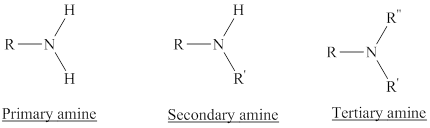
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
The boiling points of aliphatic amines are higher than those of
The
Hydrogen bonding in amine is weaker than that of alcohols or carboxylic acids. Boiling points of amines are lower than those corresponding alcohols or carboxylic acids.
Increasing order of boiling point of some organic compounds with similar molecular weight can be represented as follows,
Hydrogen bond is an attractive force established between hydrogen atom attached to a highly electronegative element and another highly electronegative element of the same or different molecule.
Intermolecular hydrogen bonding:
When hydrogen bonding takes place between various molecules of the same or different compounds, it is called intermolecular hydrogen bonding.
Molecules which have this type of hydrogen bonding possess abnormally high melting and boiling points.
Intramolecular hydrogen bonding:
When hydrogen bonding occurs within the same molecule is termed as intramolecular hydrogen bonding.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fund. of General, Org... -Masteringchem.
- C6h1206 is the chemical formula for aarrow_forwardWhy would a crystal of NaCl dissolve in water?arrow_forwardWhat product is formed when a solution of A and B is treated with mild base? This reaction is the first step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forward
- If you have a sugar with a molecular weight of 315 g/mol, how many grams would you need to add to 487 mL of water to create a 262 mM solution?arrow_forwardHow many grams of glucose (C6H2O6 molecular mass =180daltons) would be present in one liter of a 1M (molar) solution of glucose?arrow_forwardIn an experiment, what happens to the calculated molar mass after waiting an hour between thawing the frozen cyclohexane and adding the unknown compound, during which time some of the cyclohexane evaporated. Will the molar mass be too large, too small, or unaffected? Explain.|arrow_forward
- The reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardFerrous sulfate is one dietary supplement used to treat iron-deficiency anemia. What are the molecular formula and molecular mass of this compound? howmany milligrams of iron are in 250 mg of ferrous sulfate?arrow_forward
- Essentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage



