
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
12th Edition
ISBN: 9781119304241
Author: Solomons
Publisher: WILEY C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 27P
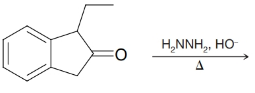
Predict the major product from each of the following reactions.
(a)
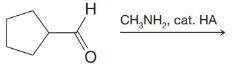
(b)
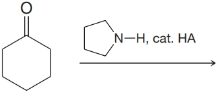
(c)
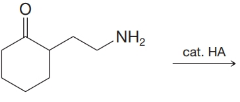
(d)
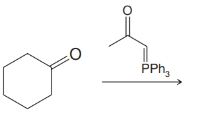
(e)
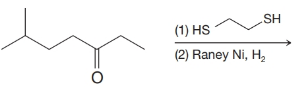
(f)
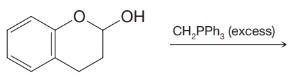
(g)
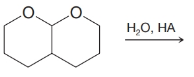
(h)
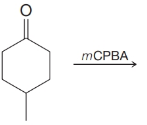
(i)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For each compound below, propose an efficient synthesis using diethyl malonate as a starting material.
Provide explanations for each step in the mechanism.
(a)
(b)
`NH2
(c)
Show how you might synthesize the following compounds, using acetylene and anysuitable alkyl halides as your starting materials. If the compound given cannot besynthesized by this method, explain why.(a) hex-1-yne (b) hex-2-yne
Show reagents to bring about each conversion.
(c)
Br
(d)
(b)
(а)
(e)
(f)
H.
Chapter 16 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Ch. 16 - PRACTICE PROBLEM 16.1 (a) Give IUPAC substitutive...Ch. 16 - Prob. 2PPCh. 16 - Prob. 3PPCh. 16 - Practice Problem 16.4
Provide the reagents and...Ch. 16 - Prob. 5PPCh. 16 - Prob. 6PPCh. 16 - Prob. 7PPCh. 16 - Prob. 8PPCh. 16 - Prob. 9PPCh. 16 - Practice Problem 16.10
Shown below is the...
Ch. 16 - Prob. 11PPCh. 16 - Practice Problem 16.12
What product would be...Ch. 16 - Prob. 13PPCh. 16 - Practice Problem 16.14
Dihydropyran reacts readily...Ch. 16 - Practice Problem 16.15 Show how you might use...Ch. 16 - Practice Problem 16.16 (a) Show how you might...Ch. 16 - Practice Problem 16.17
In addition to...Ch. 16 - Practice Problem 16.18
Triphenylphosphine can be...Ch. 16 - Prob. 19PPCh. 16 - PRACTICE PROBLEM 16.20
Give the structure of the...Ch. 16 - PRACTICE PROBLEM 16.21 What would be the major...Ch. 16 - Prob. 22PCh. 16 - 16.23 Write structural formulas for the products...Ch. 16 - Give structural formulas for the products formed...Ch. 16 - 16.25 What products would be obtained when...Ch. 16 - Predict the major organic product from each of the...Ch. 16 - 16.27 Predict the major product from each of the...Ch. 16 - 16.28 Predict the major product from each of the...Ch. 16 - Prob. 29PCh. 16 - 16.30 Write detailed mechanisms for each of the...Ch. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Show how you would convert benzaldehyde into each...Ch. 16 - 16.34 Show how ethyl phenyl ketone could be...Ch. 16 - Show how benzaldehyde could be synthesized from...Ch. 16 - Give structures for compounds AE. Cyclohexanol...Ch. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - 16.43 The structure of the sex pheromone...Ch. 16 - Provide reagents that would accomplish each of the...Ch. 16 - Write a detailed mechanism for the following...Ch. 16 - Prob. 46PCh. 16 - Dutch elm disease is caused by a fungus...Ch. 16 - Prob. 48PCh. 16 - Compounds W and X are isomers; they have the...Ch. 16 - Compounds Y and Z are isomers with the molecular...Ch. 16 - Compound A (C9H18O) forms a phenylhydrazone, but...Ch. 16 - Compound B (C8H12O2) shows a strong carbonyl...Ch. 16 - Prob. 53PCh. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - (a) What would be the frequencies of the two...Ch. 16 - Prob. 57PCh. 16 - Prob. LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
a. How does increasing the temperature increase the rate of a chemical reaction? b. How does increasing the amo...
General, Organic, and Biological Chemistry (3rd Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
11.57 Draw the cis and trans isomers for each of the following: (11.6)
a. 2-pentene
b. 3-hexene
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
The NF bond is more polar than the NH bond: but NF3 has a smaller dipole moment than NH3. Explain this curious ...
Organic Chemistry (9th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the products when each compound undergo Ozonolysis (a) (b) (c) , (d) (e)arrow_forward3) Propose suitable reagent(s) to accomplish the following transformations. (a) (b) Br N OH orarrow_forwardShow how you would synthesize octanal from each compound. You may use any necessary reagents.(a) octan-1-olarrow_forward
- (c) Answer each of the questions below that relate to acetophenone: Xo (i) (ii) (iii) Draw the structure of the enol form of acetophenone. Give a stepwise mechanism for the conversion of acetophenone into its enol form. Show how each of the three compounds A, B and C below can be prepared from acetophenone. Explain clearly what reactants/reagents would be required in each case. odocor A B Br Carrow_forwardWhich of the following is the major product of the reaction below? (1) LDA/ether (2) (3) ethanol, A (A) (B) (C) (D) OCompound A Compound D Compound B Compound Carrow_forwardProvide the reagents required to complete the following transformations. (a) □ (b) на öd io: HỌ: 0.arrow_forward
- Predict the major products of the following reactions, including stereochemistry where appropriate. (a) potassium tert-butoxide + methyl iodide (b) sodium methoxide + tert-butyl iodidearrow_forwardPredict the major products of the following reactions, and propose mechanisms tosupport your predictions.(a) pent-1-ene + HClarrow_forwardPredict the major product when each reagent reacts with ethylene oxide. (a) KCN (potassium cyanide) (b) NaN3 (sodium azide)arrow_forward
- Predict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents.(a) PBr3arrow_forwardPropose mechanisms consistent with the following reactions. (a) (b) (c) (d) (f) (breif Written explantion for each PLEASE) Propose mechanisms consistent with the following reactions. (a) HBr ROOR Br H₂SO4 (b) H₂O OH Br HBr Br (d) (c) CHBr Br NaOH Br Br (e) HCI CH3CH₂OH Br OCH2CH3 Br ED-8 CH2CH3 H- OH H+, H₂O (f) H OH CH3CH2 CH2CH3 CH2CH3 (continued)arrow_forward4) Propose suitable reagent(s) to accomplish the following transformations. (a) (b) Br. OCH3 OCH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY