
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
12th Edition
ISBN: 9781119304241
Author: Solomons
Publisher: WILEY C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 10PP
Practice Problem 16.10
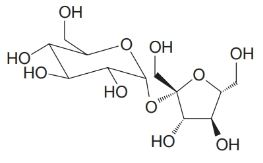
Shown below is the structural formula for sucrose (table sugar). Sucrose has two acetal groupings. Identify these.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Unknown ( C) is colorless liquid,
has 3 carbons, gives positive
result with iodoform and Lucas
tests. The oxidation of (C) formed
compound (D) which gives
negative results with Tollens, and
Jones tests. The compound (C
and D) are:
2-butanol, 2-butanone
2-pentanol and 2-pentanone
2-propanol and acetone
2-hexanol and 2- hexanone
[ 16 ] which of following compound(s) is/are neither acetal nor hemiacetal ?
он
Но
Он
осH-CHз
CHз
ОН
OCH3
(1)
( II )
( III )
(IV )
(V)
(a )
compounds III and V
(b)
compounds Il and II
(c)
compounds I and V
(d )
compounds IV and V
(e)
compounds I and IV
For each pair of compounds, predict which compound has a higher boiling point. then explain why that compound has a higher boiling point.
(a) isopropyl bromide and n-butyl bromide
(b) isopropyl chloride and tert-butyl bromide
(c) 1-bromobutane and 1-chlorobutane
Chapter 16 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Ch. 16 - PRACTICE PROBLEM 16.1 (a) Give IUPAC substitutive...Ch. 16 - Prob. 2PPCh. 16 - Prob. 3PPCh. 16 - Practice Problem 16.4
Provide the reagents and...Ch. 16 - Prob. 5PPCh. 16 - Prob. 6PPCh. 16 - Prob. 7PPCh. 16 - Prob. 8PPCh. 16 - Prob. 9PPCh. 16 - Practice Problem 16.10
Shown below is the...
Ch. 16 - Prob. 11PPCh. 16 - Practice Problem 16.12
What product would be...Ch. 16 - Prob. 13PPCh. 16 - Practice Problem 16.14
Dihydropyran reacts readily...Ch. 16 - Practice Problem 16.15 Show how you might use...Ch. 16 - Practice Problem 16.16 (a) Show how you might...Ch. 16 - Practice Problem 16.17
In addition to...Ch. 16 - Practice Problem 16.18
Triphenylphosphine can be...Ch. 16 - Prob. 19PPCh. 16 - PRACTICE PROBLEM 16.20
Give the structure of the...Ch. 16 - PRACTICE PROBLEM 16.21 What would be the major...Ch. 16 - Prob. 22PCh. 16 - 16.23 Write structural formulas for the products...Ch. 16 - Give structural formulas for the products formed...Ch. 16 - 16.25 What products would be obtained when...Ch. 16 - Predict the major organic product from each of the...Ch. 16 - 16.27 Predict the major product from each of the...Ch. 16 - 16.28 Predict the major product from each of the...Ch. 16 - Prob. 29PCh. 16 - 16.30 Write detailed mechanisms for each of the...Ch. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Show how you would convert benzaldehyde into each...Ch. 16 - 16.34 Show how ethyl phenyl ketone could be...Ch. 16 - Show how benzaldehyde could be synthesized from...Ch. 16 - Give structures for compounds AE. Cyclohexanol...Ch. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - 16.43 The structure of the sex pheromone...Ch. 16 - Provide reagents that would accomplish each of the...Ch. 16 - Write a detailed mechanism for the following...Ch. 16 - Prob. 46PCh. 16 - Dutch elm disease is caused by a fungus...Ch. 16 - Prob. 48PCh. 16 - Compounds W and X are isomers; they have the...Ch. 16 - Compounds Y and Z are isomers with the molecular...Ch. 16 - Compound A (C9H18O) forms a phenylhydrazone, but...Ch. 16 - Compound B (C8H12O2) shows a strong carbonyl...Ch. 16 - Prob. 53PCh. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - (a) What would be the frequencies of the two...Ch. 16 - Prob. 57PCh. 16 - Prob. LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Rank the given solvents in decreasing order of their ability to dissolve each compound. Solutes
Organic Chemistry (9th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Identify the reagents you would use to achieve each of the following transformations:
Organic Chemistry As a Second Language: Second Semester Topics
Draw the mechanism for the hydroxide ion-catalyzed cleavage of fructose-l.6-bisphosphate.
Organic Chemistry (8th Edition)
Predict whether a precipitation reaction will occur when aqueous solutions of the following substances are mixe...
CHEMISTRY-TEXT
Air at p=1atm enters a thin-walled ( D=5-mm diameter) long tube (L=2m) at an inlet temperature of Tm.i=100C . A...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict which member of each pair is more acidic, and explain the reasons for your predictions.(a) cyclopentanol or 3-chlorophenolarrow_forwardDraw each molecule given its name and the followinginformation. (a) Nitroglycerin, also known as 1,2,3-trinitroxypropane, the active ingredient in dynamiteand a medication administered to people having a heartattack, (Hint: The nitroxy group is the conjugate base ofnitric acid.) (b) Putrescine, also known as 1,4-diaminobutane,the compound responsible for the odor of putrefyingfish, (c) Cyclohexanone, the precursor to Nylon,(d) 1,1,2,2-tetrafluoroethene, the precursor to Teflon,(e) Oleic acid, also known as cis-9-octanedecenoic acid, amonounsaturated fatty acid found in many fats and oils.Draw the correct isomer.arrow_forwardDraw structural formulas for the hemiacetal and acetal formed from the reagents. The stoichiometry of reaction is given in the problem.arrow_forward
- Draw structural formulas for the lettered compounds (A) and (B) in the following synthetic sequence. КОН heat (A)CH16O3 (B)CH402 CHOHarrow_forwardGive a clear handwritten answer with explanation needed.. Explain the relative polarity of the three compounds based on the functional groups present: 1-Indanone, 3,4-dimethoxybenzaldehyde and 2-(3,4-Dimethoxybenzylidene)-1-indanonearrow_forwardThe odor of ripe bananas and many other fruits is due to the presence of esters. For example: Banana oil (isopentyl acetate) (a) Write the name (common or IUPAC) of the ester responsible for the fragrance of the following: pineapple, orange, apple, peach, & lavender (b) Choose one fragrant from (a) and name the alcohol and the carboxylic acid needed to synthesize this ester. (c) Show the detailed mechanism of the Fischer Esterification reaction that will be involved in the synthesis of the fragrant you have chosen in part (a).arrow_forward
- Draw the structure of sodium tetradecyl sulfate and ciprofloxacin and state how the activity of the drug is influenced by alkane groupings.arrow_forwardDraw the molecular structure of p-nitrophenol in an acidic solution and in an alkaline solution.arrow_forwardIndustrial Chemistry, Benzene is a high-commodity aromatic material with many industrial applications but is produced in relatively small amounts through petroleum fractionation. Explain how Toluene can be converted to benzene, please be specific in terms of reagents and conditions.arrow_forward
- Write an example reaction to each of the special named reactions below. a) Fischer Reaction b) Schotten-Baumann reaction c) Transesterification reactionarrow_forward6. Describe concisely a chemical test to distinguish between the following pairs of compounds. (a) n-pentanol and 3-methylpentan-3-ol (b) Ethanal dan pentanal (c) Phenol and benzoic acidarrow_forwardNaNH2 is typically used instead of KOH to produce alkynes through elimination because NaNH2 is a stronger base. Explain why KOH can be used to create diphenylacetylene.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY