
Concept explainers
Compound A
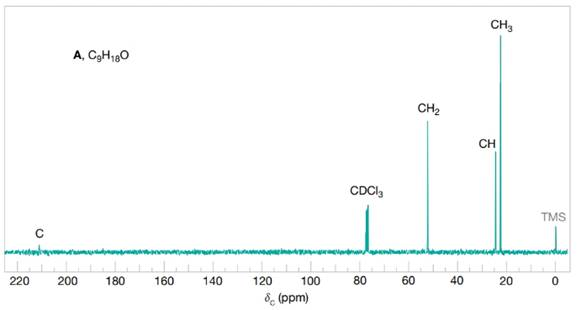
FIGURE 16.6 The broadband proton-decoupled
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Additional Science Textbook Solutions
Chemistry: A Molecular Approach (4th Edition)
Principles of Chemistry: A Molecular Approach (3rd Edition)
Living by Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
Living By Chemistry: First Edition Textbook
Chemistry: The Central Science (13th Edition)
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardA compound of formula C11H16N2 gives the IR, 1H NMR, and 13C NMR spectra shown. The proton NMR peak at δ 2 disappears on shaking with D2 Propose a structure for this compound, and show how your structure accounts for the observed absorptions.arrow_forwardThere are several isomeric alcohols and ethers of molecular formula C5H12O. Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.92 (t, 7.8 Hz, 3 H), 1.20 (s, 6H), 1.49 (q, 7.8 Hz, 2H), 1.85 (s, 1H) ppm Isomer B: δ = 1.19 (s, 9 H), 3.21 (s, 3H) ppmarrow_forward
- Shown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that reacts with a ketone in a Wittig reaction to form acompound with molecular formula C11H14. What product is obtained from the Wittig reaction?arrow_forwardThe infrared spectrum of compound A, C,H,0, shows a strong, sharp peak at 1724 cm-1. From this information and its 'H-NMR spectrum, deduce the structure of compound A. CH120 Compound A ЗН 3H 3H IH 2H 10 9. 8. 7 6. 4 3 1 O ppm Chemical shift (8)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
