
Concept explainers
(a)
The amount of height will rise the piston when the temperature is raised to
(a)
Answer to Problem 73P
The amount of height will rise the piston when the temperature is raised to
Explanation of Solution
Consider the Figure shown below.
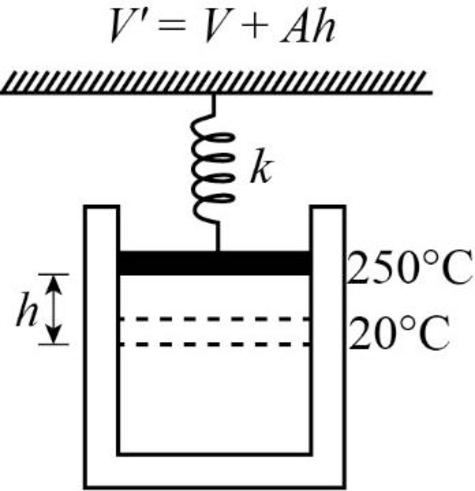
Write the ideal gas equation for the initial and final state of the cylinder when the amount of gas remains constant.
Here,
When the piston compresses the spring by a height
Use
Conclusion:
Substitute,
Solve the above equation for positive root only.
Therefore, the amount of height will rise the piston when the temperature is raised to
(b)
The pressure of the gas at
(b)
Answer to Problem 73P
The pressure of the gas at
Explanation of Solution
Conclusion:
Substitute,
Therefore, the pressure of the gas at
Want to see more full solutions like this?
Chapter 16 Solutions
Principles of Physics: A Calculus-Based Text
- A cylinder that has a 40.0-cm radius and is 50.0 cm deep is filled with air at 20.0C and 1.00 atm (Fig. P10.74a). A 20.0-kg piston is now lowered into the cylinder, compressing the air trapped inside as it takes equilibrium height hi (Fig. P16.74b). Finally, a 25.0-kg dog stands on the piston, further compressing the air, which remains at 20C (Fig. P16.74c). (a) How far down (h) does the piston move when the dog steps onto it? (b) To what temperature should the gas be warmed to raise the piston and dog back to hi?arrow_forwardA 40.0-g projectile is launched by the expansion of hot gas in an arrangement shown in Figure P12.4a. The cross sectional area of the launch tube is 1.0 cm2, and the length that the projectile travels down the tube after starting from rest is 52 cm. As the gas expands, the pressure varies as shown in Figure P12.4b. The values for the initial pressure and volume are P1 = 11 105 Pa and Vi = 8.0 cm3 while the final values are Pf = 1.0 105 Pa and Vf = 8.0 cm3. Friction between the projectile and the launch tube is negligible, (a) If the projectile is launched into a vacuum, what is the speed of the projectile as it leaves the launch tube? (b) If instead the projectile is launched into air at a pressure of 1.0 105 Pa. what fraction of the work done by the expanding gas in the tube is spent by the projectile pushing air out of the way as it proceeds down tile tube?arrow_forwardA vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P16.56). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find die height h in Figure P16.56. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder.arrow_forward
- A 40.0-g projectile is launched by the expansion of hot gas in an arrangement shown in Figure P12.4a. The cross-sectional area of the launch tube is 1.0 cm2, and the length that the projectile travels down the tube after starting from rest is 32 cm. As the gas expands, the pressure varies as shown in Figure P12.4b. The values for the initial pressure and volume are Pi = 11 x 105 Pa and Vi = 8.0 cm3 while the final values arePf = 1.0 x 105 Pa and Vf = 40.0 cm3. Friction between the projectile and the launch tube is negligible. (a) If the projectile is launched into a vacuum, what is the speed of the projectile as it leaves the launch tube? (b) If instead the projectile is launched into air at a pressure of 1.0 x 105 Pa, what fraction of the work done by the expanding gas in the tube is spent by the projectile pushing air out of the way as it proceeds down the tube?arrow_forwardThe drawing shows an ideal gas confined to a cylinder by a massless piston that is attached to an ideal spring. Outside the cylinder is a vacuum. The cross-sectional area of the piston is A=2.5 x 10^-3 m^2. The initial pressure, volume, and temperature of the gas are, respectively, p0, v0= 6.00 x 10^-4 m^3 and t0=273 K , the spring is initially stretched by 0.095 m with respect to its unstrained length. The gas is heated, so that its final pressure, volume, and temperature are Pf, Vf, and Tf and the spring is stretched by an amount of 0.12 meters with respect to its unstrained length. What is the final temperature of the gas?arrow_forwardThe density of helium gas at 0°C is ρ0 = 0.179 kg/m3. The temperature is then raised to T = 170°C, but the pressure is kept constant. Assuming that helium is an ideal gas, calculate the new density ρf of the gas. kg/m3arrow_forward
- A clown at a birthday party has brought along a helium cylinder, with which he intends to fill balloons. When full, each balloon contains 0.00440 m3 of helium at an absolute pressure of 1.10 x 105 Pa. The cylinder contains helium at an absolute pressure of 1.90 x 107 Pa and has a volume of 0.00320 m3. The temperature of the helium in the tank and in the balloons is the same and remains constant. What is the maximum number of people who will get a balloon?arrow_forwardIf during an expansion process the volume of a gas changes from 0.2 to 0.5 m^3 and pressure changes according to equation P= 1500(v/100+1) where P is in Pa and V is in m^3. What is the work done by gas in kJ?arrow_forwardThe internal air pressure of a fridge reduces after closing its door due to cooling of air. If the temperature of all the trapped air falls from 30 deg Celcius to 5.0 deg Celcius after closing the fridge door, what is the magnitude of the resulting net force on the door? Take the volume of trapped air to be 0.30 m3, take the fridge to be perfectly sealed, and take the door area that is in contact with the trapped air to be 0.60 m2?arrow_forward
- A student foolishly attempts to stop a steel bar, of length L = 1 m and at a temperature of 20ºC, from thermally expanding by attaching it to a wooden support with a nail at each end. Steel's Young's modulus is Y = 1.1 × 1011 N/m2 and it's linear thermal expansion coefficient is α = 13 × 10-6 1/C. Randomized Variables Y = 1.1 × 1011 N/m2α = 13 × 10-6 1/C What is the volue of the stress, in pascals, that develops due to a rise of temprature to 21 C? Assuming the nails have a cross- sectional area of A= 10^-5 m^2 all of which is perpendicular to the stress force from the bar, what is the force acting on each due to that temperature rise?arrow_forwardAn ideal gas is confined in a container whose volume is fixed. If the container holds n moles of gas, by what factor will the pressure increase if the absolute temperature is increased by a factor of 2? (a) 2 / (nR) (b) 2 (c) 2nR (d) 2 / n (e) 2 / Rarrow_forwardA steel rod and an aluminum rod are originally the same length, 50cm, at -8 degrees C. What is the difference in the lengths of the two rods at 110 degrees C? The coefficients of linear expansion are 1.2 x 10-5 K-1 for steel and 2.4 x 10-5 K-1 for aluminum. a).7 mm b)1.4 mm c)5.2 mm d)2.1 mmarrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

