
Organic Chemistry, Books A La Carte Edition (8th Edition)
8th Edition
ISBN: 9780321777690
Author: Leroy G. Wade
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 95P
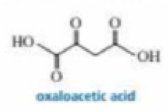
The pKa values of the carboxylic acid groups of oxaloacetic acid are 2.22 and 3.98.
- a. Which carboxyl group is the stronger acid?
- b. The amount of hydrate present in an aqueous solution of oxaloacetic acid depends on the pH of the solution: 95% at pH 0, 81% at pH 1.3, 35% at pH 3.1, 13% at pH 4.7, 6% at pH 6.7, and 6% at pH 12.7. Explain this pH dependence.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The pKa values of the carboxylic acid groups of oxaloacetic acid are 2.22 and 3.98.a. Which carboxyl group is the stronger acid?b. The amount of hydrate present in an aqueous solution of oxaloacetic acid depends on the pH of the solution: 95% at pH 0, 81% at pH 1.3, 35% at pH3.1, 13% at pH 4.7, 6% at pH 6.7, and 6% at pH 12.7. Explain this pH dependence.
The pKa values of the carboxylic acid groups of oxaloacetic acid are 2.22 and 3.98.
a. Which carboxyl group is the stronger acid? b. The amount of hydrate present in an aqueous solution of oxaloacetic acid depends on the pH of the solution: 95% at pH 0, 81% at pH 1.3, 35% at pH 3.1, 13% at pH 4.7, 6% at pH 6.7, and 6% at pH 12.7. Explain this pH dependence.
Which has a lower pKa value, the conjugate acid of 3-bromoquinuclidine or the conjugate acid of 3 -chloroquinuclidine?
Chapter 16 Solutions
Organic Chemistry, Books A La Carte Edition (8th Edition)
Ch. 16.1 - Give two names for each of the following:Ch. 16.1 - Prob. 2PCh. 16.1 - Name the following:Ch. 16.2 - Prob. 4PCh. 16.4 - What products are formed when the following...Ch. 16.4 - We saw on the previous page that...Ch. 16.4 - a. How many stereoisomers are obtained from the...Ch. 16.4 - Prob. 9PCh. 16.4 - Write the mechanism for the reaction of acetyl...Ch. 16.4 - Prob. 11P
Ch. 16.4 - Show how the following compounds can be...Ch. 16.4 - Prob. 13PCh. 16.4 - Prob. 14PCh. 16.4 - In the mechanism for cyanohydrin formation, why is...Ch. 16.4 - Prob. 16PCh. 16.4 - Prob. 17PCh. 16.4 - Show two ways to convert an alkyl halide into a...Ch. 16.5 - Prob. 20PCh. 16.5 - Prob. 21PCh. 16.5 - Prob. 22PCh. 16.5 - Prob. 23PCh. 16.6 - Prob. 24PCh. 16.7 - What reducing agents should be used to obtain the...Ch. 16.7 - Prob. 26PCh. 16.8 - Prob. 27PCh. 16.8 - Prob. 28PCh. 16.8 - Prob. 29PCh. 16.8 - The pKa of protonated acetone is about 7.5. and...Ch. 16.8 - Prob. 31PCh. 16.8 - Prob. 32PCh. 16.8 - Prob. 33PCh. 16.8 - Excess ammonia must be used when a primary amine...Ch. 16.8 - The compounds commonly known as amino acids are...Ch. 16.9 - Hydration of an aldehyde is also catalyzed by...Ch. 16.9 - Which ketone forms the most hydrate in an aqueous...Ch. 16.9 - When trichloroacetaldehyde is dissolved in water,...Ch. 16.9 - Which of the following are a. hermiacetals? b....Ch. 16.9 - Prob. 40PCh. 16.9 - Explain why an acetal can be isolated but most...Ch. 16.10 - Prob. 42PCh. 16.10 - Prob. 43PCh. 16.10 - What products would be formed from the proceedings...Ch. 16.10 - a. In a six-step synthesis, what is the yield of...Ch. 16.10 - Show how each of the following compounds could be...Ch. 16.12 - Prob. 47PCh. 16.13 - Prob. 49PCh. 16.14 - Prob. 50PCh. 16.15 - Prob. 51PCh. 16.16 - Prob. 52PCh. 16 - Draw the structure for each of the following: a....Ch. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - a. Show the reagents required to form the primary...Ch. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - Using cyclohexanone as the starting material,...Ch. 16 - Propose a mechanism for each of the following...Ch. 16 - Show how each of the following compounds can be...Ch. 16 - Fill in the boxes:Ch. 16 - Prob. 64PCh. 16 - Identify A through O:Ch. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - How many signals would the product of the...Ch. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - Prob. 74PCh. 16 - Prob. 75PCh. 16 - Prob. 76PCh. 16 - Prob. 77PCh. 16 - Prob. 78PCh. 16 - Draw structure for A-D for each of the following:Ch. 16 - Prob. 80PCh. 16 - a. Propose a mechanism for the following reaction:...Ch. 16 - Prob. 82PCh. 16 - A compound gives the following IR spectrum. Upon...Ch. 16 - How can be following compounds be prepared from...Ch. 16 - Prob. 85PCh. 16 - Prob. 86PCh. 16 - Prob. 87PCh. 16 - In the presence of an acid catalyst, acetaldehyde...Ch. 16 - Prob. 89PCh. 16 - Prob. 90PCh. 16 - Prob. 91PCh. 16 - A compound reacts with methylmagnesium bromide...Ch. 16 - Show how each of the following compounds can be...Ch. 16 - Prob. 94PCh. 16 - The pKa values of the carboxylic acid groups of...Ch. 16 - The Baylis-Hillman reaction is a DABCO...Ch. 16 - Prob. 97PCh. 16 - Prob. 98P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The pKa values of a few ortho-, meta-, and para-substituted benzoic acids are shown below: The relative pKa values depend on the substituent. For chloro-substituted benzoic acids, the ortho isomer is the most acidic and the para isomer is the least acidic; for nitro-substituted benzoic acids, the ortho isomer is the most acidic and the meta isomer is the least acidic; and for amino-substituted benzoic acids, the meta isomer is the most acidic and the ortho isomer is the least acidic. Explain these relative acidities. a. Cl: ortho > meta > para b. NO2: ortho > para > meta c. NH2: meta > para > orthoarrow_forwardThe pKa values of a few ortho-, meta-, and para-substituted benzoic acids are shown below: The relative pKa values depend on the substituent. For chloro-substituted benzoic acids, the ortho isomer is the most acidic and the para isomer is the least acidic; for nitro-substituted benzoic acids, the ortho isomer is the most acidic and the meta isomer is the least acidic; and for amino-substituted benzoic acids, the meta isomer is the most acidic and the ortho isomer is the least acidic. Explain these relative acidities.a. Cl: ortho 7 meta 7 para b. NO2: ortho 7 para 7 meta c. NH2: meta 7 para 7 orthoarrow_forwarda. For this acid-base reaction: C⊖H3 + H3O+ −−→ CH4 + H2O, the conjugate acid of carboanion (C⊖H3) is _________________ b. Complete the equation: HC≡N + NaOH −−→ ___________+__________ c. Rank the acidity of these: NH3, HF, H2O, HCl, NaH Most acidic______>_______>_______>_______>_______least acidic d. The Ka of acetic acid (found in vinega) is 1.8×10-5. Then the pKa of formic acid isarrow_forward
- Dicarboxylic acids have two pKa's.- For maleic acid (cis-2-butenedioic acid) these are pKa1 = 2.0, and pKa2 = 6.3- For fumaric acid (trans-2-butenedioic acid) these are pKa1 = 3.0, and pKa2 = 4.5 Which factor best explains why the cis-isomer has a smaller pKa1 and a larger pKa2 than the trans-isomer? a. Intramolecular hydrogen bonding b. Intramolecular steric hindrance c. Selective solvation in water d. Intramolecular dipole repulsionarrow_forwardPhthalic acid and isophthalic acid have protons on two carboxy groups that can be removed with base. (a) Explain why the pKa for loss of the first proton (pKa1) is lower for phthalic acid than isophthalic acid. (b) Explain why the pKa for loss of the second proton (pKa2) is higher for phthalic acid than isophthalic acid.arrow_forwardWhat percent of acetic acid is present in the acidic form at pH 5.0, assuming a pKa of 4.8?arrow_forward
- Which carboxylic acid has the lower pKa, pyruvic acid (CH3COCO2H) or acetoacetic acid (CH3COCH2CO2H)? Explain your choice.arrow_forwardBenzoic acid (C6H5C02H) has Ka =6.3 x 10-5 a) What is the value of Kb for the benzoate ion (C6H5C02-)? b) What is the h30+ concentration of a O, 10 mol/dm3 solution of benzoic acid? c) What is the pH of the solution in (b) above?arrow_forward3. If you have an unknown carboxylic acid, what pH you need to achieve upon re-acidification with HCl to ensure that your carboxylic acid is 99.9% back in its charge neutral form?Assume a pKa of 5 for your carboxylic acid, and remember that the pH needs to be 3 units different from the pKa for 99.9% ionization or charge neutralization. pH 7 or lower pH 5 or lower pH 2 or lower pH 13 or lowerarrow_forward
- When water is the solvent, the pKa of acetic acid (CH3CO2H) is 4.75, but when DMSO is the solvent, the pKa is 12.6.Explain why. Hint: Review Section 2.9 and consider the ability of each solvent to solvate cations and anions.arrow_forwardSuppose you have a mixture of these three compounds. Devise a chemical procedure based on their relative acidity or basicity to separate and isolate each in pure form.arrow_forwardDicarboxylic acids have two dissociation constants, one for the initial dissociation into a monoanion and one for the second dissociation into a dianion. For oxalic acid, HO2C—CO2H, the first ionization constant is pKal = 1.2 and the second ionization constant is pKa2 = 4.2. Why is the second carboxyl group far less acidic than the first?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License